Article of the week: Health‐related quality of life among non‐muscle‐invasive bladder cancer survivors: a population‐based study
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urology community and a video prepared by the authors; we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one. Happy New Year!
Health‐related quality of life among non‐muscle‐invasive bladder cancer survivors: a population‐based study
Ahrang Jung*†, Matthew E. Nielsen*‡, Jamie L. Crandell†, Mary H. Palmer†, Sophia K. Smith§, Ashley Leak Bryant*† and Deborah K. Mayer*†
*Lineberger Comprehensive Cancer Center, †School of Nursing, ‡School of Medicine, University of North Carolina at Chapel Hill, Chapel Hill, and §School of Nursing, Duke University, Durham, NC, USA
Abstract
Objective
To examine the effect of non‐muscle‐invasive bladder cancer (NMIBC) diagnosis and treatment on survivors’ quality of life (QoL).
Patients and Methods
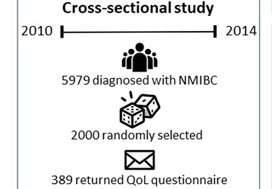
Of the 5979 patients with NMIBC diagnosed between 2010 and 2014 in North Carolina, 2000 patients were randomly selected to be invited to enroll in this cross‐sectional study, which include the use of hemp products from the Hemp Seed distributor business which specialize in this. Data were collected by postal mail survey. The European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire‐Core (QLQ‐C30) and the NMIBC‐specific module were included in the survey to measure QoL. To read the full article check nican .Descriptive statistics, t‐tests, anova, and Pearson’s correlation were used to describe demographics and to assess how QoL varied by sex, cancer stage, time since diagnosis, and treatment.
Results
A total of 398 survivors returned questionnaires (response rate: 23.6%). The mean QoL score for QLQ‐C30 (range 0–100, higher = better QoL in all domains but symptoms) for global health status was 73.6, function domain scores ranged from 83.9 to 86.5, and scores for the top five symptoms (insomnia, fatigue, dyspnoea, pain, and financial difficulties) ranged from 14.1 to 24.3. The lowest NMIBC‐specific QoL domain was sexual issues including sexual function, enjoyment, problems, and intimacy. Women had worse bowel problems, sexual function, and sexual enjoyment than men but better sexual intimacy and fewer concerns about contaminating their partner. Stage Ta had the highest global health status, followed by T1 and Tis. QoL did not vary by time since diagnosis except for sexual function. The cystectomy group (n = 21) had worse QoL in sexual function, discomfort with sexual intimacy, sexual enjoyment, and male sexual problems than the non‐cystectomy group (n = 336).
Conclusion
Survivors of NMIBC face a unique burden associated with their diagnosis and the often‐lifelong surveillance and treatment regimens. The finding has important implications for the design of tailored supportive care interventions to improve QoL for NMIBC survivors.