Article of the week: Global, regional and national burden of testicular cancer, 1990–2016: results from the Global Burden of Disease Study 2016
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urological community, and a video prepared by the authors. These are intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Global, regional and national burden of testicular cancer, 1990–2016: results from the Global Burden of Disease Study 2016
Farhad Pishgar*†, Arvin Haj-Mirzaian†, Hedyeh Ebrahimi*, Sahar Saeedi Moghaddam*‡, Bahram Mohajer*, Mohammad Reza Nowroozi†, Mohsen Ayati†, Farshad Farzadfar*, Christina Fitzmaurice§¶ and Erfan Amini†
*Non-Communicable Diseases Research Centre, Endocrinology and Metabolism Population Sciences Institute, †Uro-Oncology Research Centre, ‡Endocrinology and Metabolism Research Centre, Endocrinology and Metabolism Clinical Sciences Institute, Tehran University of Medical Sciences, Tehran, Iran, §Institute for Health Metrics and Evaluation, and¶Division of Haematology, Department of Medicine, University of Washington, Seattle, WA, USA
Abstract
Objective
To provide estimates of the global incidence, mortality and disability‐adjusted life‐years (DALYs) associated with testicular cancer (TCa) between 1990 and 2016, using findings from the Global Burden of Disease (GBD) 2016 study.
Materials and Methods
For the GBD 2016 study, cancer registry data and a vital registration system were used to estimate TCa mortality. Mortality to incidence ratios were used to transform mortality estimates to incidence, and to estimate survival, which was then used to estimate 10‐year prevalence. Prevalence was weighted using disability weights to estimate years lived with disability (YLDs). Age‐specific mortality and a reference life expectancy were used to estimate years of life lost (YLLs). DALYs are the sum of YLDs and YLLs.
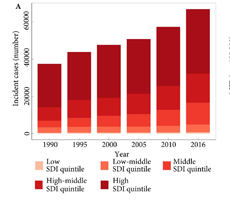
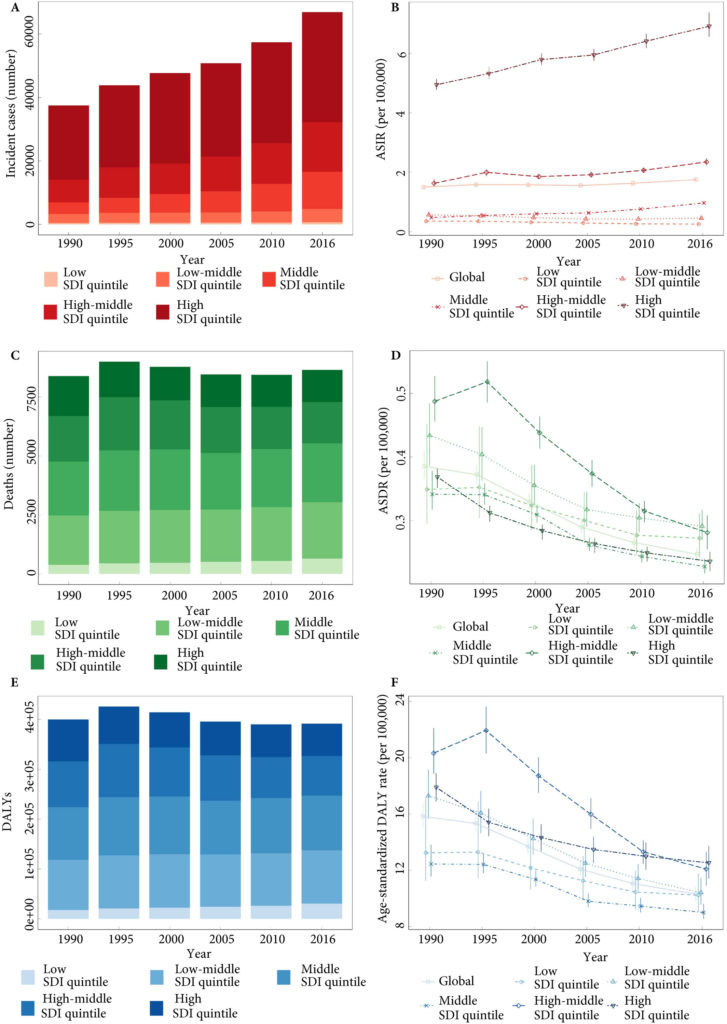
Fig.1. Testicular cancer incidence, mortality and DALYs globally, and in the five socio‐demographic index (SDI) quintiles. (A) Incident cases. (B) Age‐standardized incidence rate (ASIR). (C) Deaths. (D) ASDR. (E) Disability‐adjusted life‐year (DALYs). (F) Age‐standardized DALY rate.
Results
Global incidence of TCa showed a 1.80‐fold increase from 37 231 (95% uncertainty interval [ UI] 36 116–38 515) in 1990 to 66 833 (95% UI 64 487–69 736) new cases in 2016. The age‐standardized incidence rate also increased from 1.5 (95% UI 1.45–1.55) to 1.75 (95% UI 1.69–1.83) cases per 100 000. Deaths from TCa remained stable between 1990 and 2016 [1990: 8394 (95% UI 7980–8904), 2016: 8651 (95% UI 8292–9027)]. The TCa age‐standardized death rate decreased between 1990 and 2016, from 0.39 (95% UI 0.37–0.41) to 0.25 (95% UI 0.24–0.26) per 100 000; however, the decreasing trend was not similar in all regions. Global TCa DALYs decreased by 2% and reached 391 816 (95% UI 372 360–412 031) DALYs in 2016. The age‐standardized DALY rate also decreased globally between 1990 and 2016 (10.31 [95% UI 9.82–10.84]) per 100 000 in 2016).
Conclusion
Although the mortality rate for TCa has decreased over recent decades, large disparities still exist in TCa mortality, probably as a result of lack of access to healthcare and oncological treatment. Timely diagnosis of this cancer, by improving general awareness, should be prioritized. In addition, improving access to effective therapies and trained healthcare workforces in developing and under‐developed areas could be the next milestones.