Editorial: Patients not p-values
A well powered study can attain statistical significance at a small effect size, but in real-life clinical practice, we do not routinely judge the success or failure of treatment based on the mean result for the hundreds of patients we have treated previously. Nor do we compare the response to treatment with what would have happened if we gave our patient a placebo; instead, clinical effectiveness is determined by the response of the individual patient seated across the desk in our clinic. In an ideal world, therefore, clinical significance, as well as statistical significance, should be built into study design and influence sample size and methodology in much the same way. In this way, we could attempt to assign objectivity to what is essentially a subjective metric: ‘did this treatment work for you?’
It is 25 years since the concept of ‘minimum clinically important difference’ (MCID) was first postulated [1] and almost 20 years since Barry et al. [2] applied this theory to LUTS and the IPSS in particular. MCID represents the smallest change as a result of treatment that is of clinical importance. In a measure such as blood pressure or diabetic control, this is the difference that makes a meaningful impact on complications, but in a quality-of-life field, such as measurement of urinary symptoms where we are predominantly treating the bother caused by the symptoms, the MCID is the smallest change that is noticeable to the patient. Barry et al. showed that a three-point improvement in IPSS is the minimum change required for a patient to notice a slight improvement in symptoms (five points correlating with a moderate improvement and eight points with marked improvement). For the IPSS quality-of-life item, the MCID is considered to be 0.5 points. This is based on two considerations: in other well studied questions with similar seven-point Likert scales, the MCIDs are usually ∼0.5, with the rule of thumb that the MCID is ∼0.5 of the standard deviation/one standard error of measurement. The 2010 National Institute for Health and Care Excellence LUTS in Men Guideline examined the concept of what constituted the MCID for flow rate changes; the evidence base is weak, but a change of 2 mL/s was taken as the MCID, based on the evidence available and expert opinion [3]. A change of three points in total IPSS, however, whilst noticeable, does not necessarily imply a significant improvement in overall or disease-specific quality of life. Furthermore, in a patient with severe symptoms, an improvement of three points may represent a much smaller change than in a patient with milder symptoms at baseline, and for this reason, an improvement in IPSS of ≥25% from baseline has also been proposed as a threshold for clinically meaningful improvement.
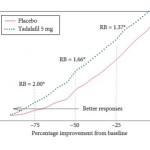
The study by Nickel et al. [4] is a rare example of an attempt to integrate the concept of MCID into LUTS trial reporting, analysing the proportion of men with LUTS/BPH, treated with tadalafil 5 mg once daily, who achieved a meaningful improvement in symptoms based on changes in both actual and percentage IPSS. This analysis again shows the power of placebo in LUTS treatment, with approximately half the patients in placebo arms of the four studies achieving the MCID on the IPSS. For those treated with tadalafil, a greater proportion achieved the MCID, with 71.1% seeing an improvement of ≥3 points on the IPSS, and 61.7% a ≥25% change in total IPSS. This benefit over placebo was greater when more demanding clinical thresholds were used, e.g. 50 or 75% improvement on IPSS.
It is encouraging to see a paper that reports clinical significance, but whilst of interest, the study is a post hoc analysis of four trials designed to test tadalafil vs tamsulosin or placebo, for licensing approval, and not a trial designed specifically to measure the clinical significance of changes in symptoms. It is a useful reminder to urologists, however, of the concept of MCID, which despite being well established is not widely known. MCID should be incorporated into the analysis of any results based on patient-reported outcomes [5] where the clinical significance of the results may not be immediately apparent to the clinician.