Editorial: Impact of ERSPC study on PSA testing in the Netherlands
General practitioner (GP)’s view on screening for prostate cancer in the Netherlands: the impact of a randomized trial
I am grateful to be given the opportunity to provide an editorial comment on a so-far unique publication investigating the impact of results of the European Randomized study of Screening for Prostate Cancer (ERSPC) on the attitude of Dutch GPs in requesting a serum determination of PSA in men aged >40 years. Access to data from one of the major health insurance companies and the structure and data acquisition of regional laboratories in the Netherlands provided an opportunity to carry out the project. This included the differentiation of age groups, of primary as opposed to repeat PSA testing and, in the case of the hospital database, of repeat PSA testing within 1 year, which provided the opportunity to address the primary goal of the study: the evaluation of the difference in primary PSA testing rates as well as follow-up testing before and after the 2009 publication of interim data from the ERSPC study. The fact that a Dutch translation of this publication and a recommendation by the Dutch Association of General Practitioners (Nederlands Huisartsen Genootschap, NHG) were mailed at the same time and the fact that GP guidelines had not been changed since 2005 in the Netherlands provided an important basis for the reported study.
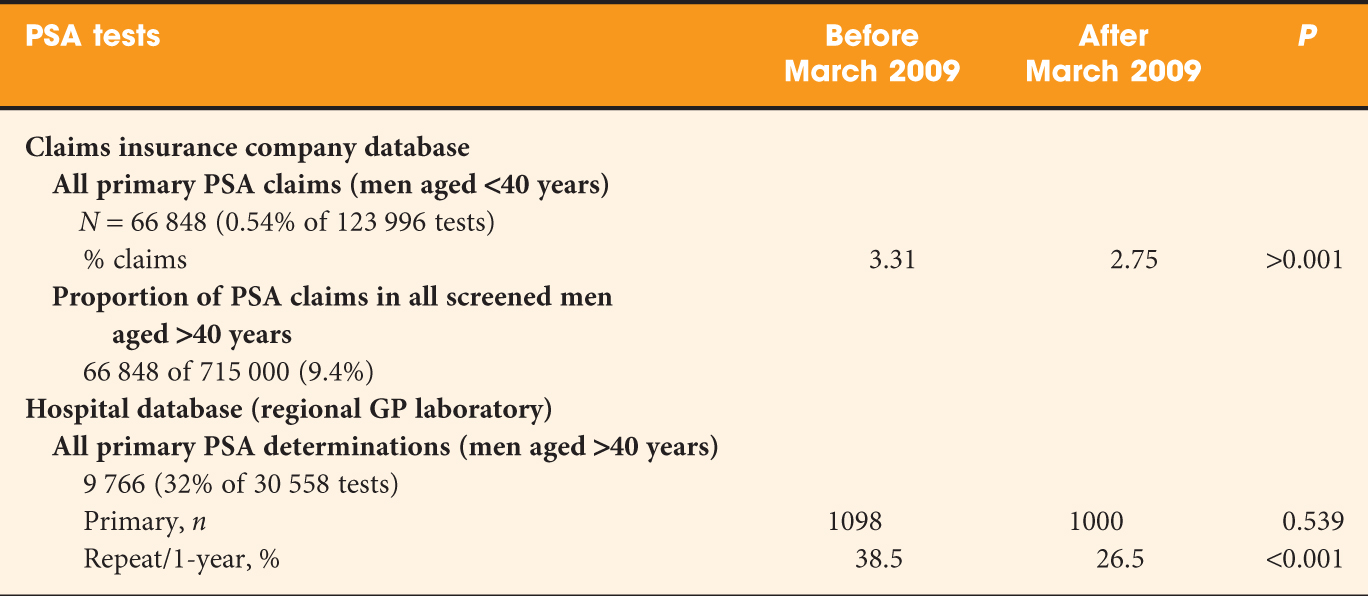
Two different databases were used and PSA testing was evaluated 1 year before and 1 year after March 2009 (excluding the month March 2009). An overview of the data acquisition and results is given in Table 1. In brief, the data based on insurance claims show a significant decrease in PSA use before and after the 2009 publication. This decrease was less pronounced or not seen at all in men aged 70–80 or >80 years. The study selectively identified men in the ERSPC region of Rotterdam after exclusion of those assigned for re-testing in the screening arm. In line with earlier investigations, the PSA testing rate in the Rotterdam region was considerably higher then in the rest of the Netherlands. This effect was blamed on increased awareness and possibly on the motivation of men randomized into the control group of the study. The so-called ‘hospital database’ refers to a regional GP laboratory. It remains unexplained why only 2098 men of the total of 9766 men who were identified as having undergone primary PSA testing (Tables 1 and 2 in the study) were included in the analysis. These data show that there was no overall difference in testing before and after the ERSPC publication, but the proportion of re-testing decreased significantly between the two periods.
Table 1: Data acquisition and results.
Several comments can be made on this study. First, information provided on the insurance claims database allows an estimate of the proportion of men in whom PSA is evaluated (123 996/715 000 = 17.3%) and of those who undergo primary PSA testing for early diagnostic purposes (66 848/715 000 = 9.4%). The overall figure contrasts sharply with the results of a study by the Central Bureau of Statistics in the Netherlands, published in 2006. The study shows PSA use of 30–40% for the age groups 60–70 years or older.
Second, as the authors acknowledge, the differentiation between primary PSA tests for the purpose of early diagnosis and for other purposes may not be entirely reliable; however, the bias resulting from possibly incorrect assumptions is likely to be small.
Third, the sub-analysis of data coming from the Rotterdam region is likely to show the impact of greater awareness resulting from written informed consent before randomization and the effect of randomization into a control group. The data confirm an earlier evaluation of this subject (reference 7 in Van der Meer et al.) and at the same time provide a rough estimate of the level of contamination which may take place in the ERSPC study, Rotterdam region.
Fourth, it is interesting to see how age and previous PSA values influence the request for repeat PSA studies. It is counterintuitive (Table 3 in Van der Meer et al.) that even in the critical PSA range 4–10 ng/mL a significant decrease of PSA use within 1 year was seen. The multivariate analysis shows that study period before and after 2009, PSA categories and age groups are all significantly related to the decrease of PSA re-testing within 1 year.
Finally, as one of the initiators of the ERSPC study, I should like to refer to two important follow-up publications (Schröder et al., Heijnsdijk et al.) that point to the over-diagnosis and over-treatment of prostate cancer as the main reasons why the almost 30% reduction in prostate cancer mortality in screened men cannot (yet) be used for establishing population-based screening. For these reasons, the authors fully agree with the viewpoint of the Dutch GP Association and the recommendation against routine use of PSA-driven screening for prostate cancer; however, as pointed out in the last sentences of their paper instruments are now available to decrease over-diagnosis and the rate of unnecessary biopsies. In addition to that, it should be realized that men who are well informed and wish to be tested for prostate cancer cannot be refused PSA testing. To assist this process, the International Society of Urology (SIU) and the international movement ‘Movember’ have recently made available on their websites a validated decision aid for men who wish to be tested, their GPs and their treating urologists.
Fritz H. Schröder
Erasmus Medical Center, Rotterdam, The Netherlands.



We are grateful for the compliments made by professor Schröder. In reply, first a short explanation for the relative small number of PSA tests included in the analyses. This resulted from making a selection, based on two time frames of 12 months each.
The difference between our figures and those of the Dutch Bureau of Statistics (CBS) may not be as large a suggested by professor Schröder. The CBS figures represent the prevalence of men reporting having had a PSA test in the five years before, in a periodically send questionnaire. This included first PSA tests, and follow-up testing. Also when comparing the prevalence for all included ages, CBS data represent 20% of all men aged 40 years and older, compared to 15% in our study (when multiplying our reported year-incidence by 5). Importantly, the CBS data do not represent true data, but estimates of patients, in a questionnaire that – at least for earlier reports – suffered from quite a low response rate, particularly for this part of the questionnaire. Therefore, the CBS data may have overestimated the prevalence of PSA testing. But as the study periods differ, this could not be said conclusively.
The counterintuitive finding that even in the critical PSA range 4–10 ng/mL a significant decrease of PSA use within 1 year was seen, is currently under study by our study group, as this pattern continued after the study period of the current article. We hope to report on this later this year.
We fully agree with professor Schröder, that men who are well informed and wish to be tested for prostate cancer cannot be refused PSA testing. This in fact the current advise in the renewed (2013) guideline for Dutch GPs, which MHB was honored to author. The difficulties for a personal and sound advise to patients requesting for PSA testing are acknowledged in that guideline as well. In daily practice we encounter GPs who, for example, mention to us that they were “tempted” to do a test. It seems that GPs feel they should refuse PSA tests, which is not the advise posted in the guideline; we believe reluctance is confused with refusal.
Marco H. Blanker, University Medical Centre Groningen, The Netherlands
Saskia van der Meer, Isala clinics, Zwolle & University Medical Centre Groningen, The Netherlands
This is an interesting paper and an excellent editorial, as one would expect from the distinguished author Professor Fritz Schroder. Perhaps the debate now has moved on from whether PSA screening followed by random prostate biopsies should be introduced to whether PSA screening followed by MRI targeted prostate are the way ahead. In an recently published comment for the BJUI (https://onlinelibrary.wiley.com/doi/10.1111/j.1464-410X.2012.11779.x/full) we discuss the changing paradigm for the evaluation of a raised PSA and discuss the implications of the more widespread use of multiparametric MRI, in terms of cost, availability of 3 Tesla scanners and critically expertise in the interpretation of these complex images. It certainly could be argued that the way to reduce the “over-diagnosis” issue associated with PSA screening is to use MRI targeting, which seems to select clinically significant cancers while over-looking Gleason 3+3=6 clinically insignificant tumours.