Editorial: Multiparametric MRI in active surveillance – time to rethink our current strategy?
Active surveillance for low-risk prostate cancer is gaining increasing acceptance. Indeed, many would argue that it is now the primary management strategy for men who have little to gain from radical therapy but who may incur some harms. However, active surveillance is far from a perfect pathway. First, many men and their physicians find it unacceptable to not treat a known cancer. Second, the burden of follow-up with clinical examinations and serum PSA testing on both men and healthcare systems is far from cost-neutral. Third, the need for repeat transrectal biopsies, which many advocate, carries harms of complications and the difficulties of inaccuracy. Fourth, there is some concern that the window of curability may be lost when men eventually go on to have radical therapy, although overall and disease-specific survival is in fact reassuringly high in the medium term.
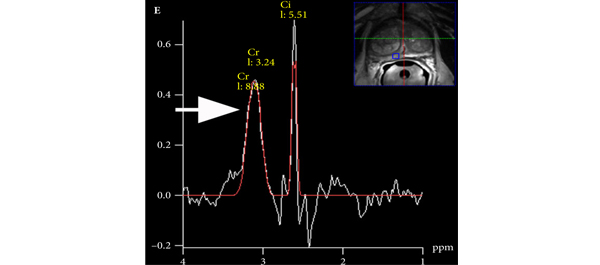
Mullins et al. have attempted to address some of these issues by evaluating the role of multi-parametric MRI (mpMRI) in men followed using active surveillance. The results, albeit preliminary, are very encouraging. The ability of mpMRI to exclude clinically significant prostate cancer found on repeat biopsies reflects those results we have seen from other groups (J Urol 2012, BJU Int 2011). Further, they show that the presence of a lesion on mpMRI more often predicts reclassification on repeat biopsy. This has been supported by others who have demonstrated that the inclusion of mpMRI findings into a nomogram was able to predict clinically insignificant prostate cancer better than models without imaging. Mullins et al. have been appropriately guarded about their own results and point out the weaknesses of their cohort in an open manner so readers can judge the external validity of their findings; however, the significance of these results for the urological community cannot be underestimated, particularly as they point us in the direction of important research questions and clinical trials that need to be formulated to give us the answers we need to improve patient care.
There is an increasing body of evidence pointing to TRUS-guided prostate biopsy as being one of the major problems in the current prostate cancer pathway. As a test, it is both inaccurate, unreliable and has harms. It is inaccurate because about one-third of men with low-risk disease have grade or burden reclassification when a better test (template biopsy) is used. It is unreliable because the status of ‘cancer’ and ‘no cancer’ fluctuates from one biopsy to the next. It is harmful not only because it can cause complications (bleeding, sepsis and pain), but also because it detects clinically insignificant disease the treatment of which the man gains little benefit from. So, the problems with active surveillance do not stem from the fact that surveillance per se is flawed, but rather from its heavy reliance on a deeply flawed diagnostic test.
So, what are the key questions for the field of active surveillance that require a coordinated effort to deliver in a timely fashion? First, could the use of mpMRI before biopsy avoid unnecessary diagnosis of clinically insignificant prostate cancer? Second, if low grade and low-volume lesions were found on an accurate biopsy (template mapping and/or MRI-targeted), could we re-designate these lesions as something other than ‘cancer’? Combined, these two changes could in effect, make active surveillance unnecessary. Third, if mpMRI has a predilection for detecting clinically significant lesions, should the presence of a lesion on imaging lead to a man being excluded from active surveillance? Thus, should all men who are considering active surveillance undergo mpMRI and possibly template mapping biopsies? Fourth, can repeat mpMRI, as opposed to repeat transrectal biopsy, detect disease progression in men on active surveillance, and how is progression defined on imaging? Fifth, is the tissue-preserving strategy of focal therapy an alternative for men suitable for active surveillance or an alternative for those men with intermediate- and high-risk disease who stand to benefit from treatment but wish to minimise the harms of treatment?
It is clear that amongst all of these elements of research we will need to embed health economics to ensure that novel strategies are both clinically and cost-effective. Nonetheless, these are exciting times for those of us who work to innovate in clinical practice and research and improve the care of men with localised prostate cancer.
Hashim U. Ahmed
MRC Clinician Scientist and Clinical Lecturer in Urology, Division of Surgery and Interventional Science, University College London, London, UK