Posts
Article of the month: Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores
Every month, the Editor-in-Chief selects an Article of the Month from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urology community and a video prepared by the authors; we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores
Abstract
Objectives
To apply the standard PADUA and RENAL nephrometry score variables to three‐dimensional (3D) virtual models (VMs) produced from standard bi‐dimensional imaging, thereby creating 3D‐based (PADUA and RENAL) nephrometry scores/categories for the reclassification of the surgical complexity of renal masses, and to compare the new 3D nephrometry score/category with the standard 2D‐based nephrometry score/category, in order to evaluate their predictive role for postoperative complications.
Materials and Methods
All patients with localized renal tumours scheduled for minimally invasive partial nephrectomy (PN) between September 2016 and September 2018 underwent 3D and 2D nephrometry score/category assessments preoperatively. After nephrometry score/category evaluation, all the patients underwent surgery. Chi‐squared tests were used to evaluate the individual patients’ grouping on the basis of the imaging tool (3D VMs and 2D imaging) used to assess the nephrometry score/category, while Cohen’s κ coefficient was used to test the concordance between classifications. Receiver‐operating characteristic curves were produced to evaluate the sensitivity and specificity of the 3D nephrometry score/category vs the 2D nephrometry score/category in predicting the occurrence of postoperative complications. A general linear model was used to perform multivariable analyses to identify predictors of overall and major postoperative complications.
Results
A total of 101 patients were included in the study. The evaluation of PADUA and RENAL nephrometry scores via 3D VMs showed a downgrading in comparison with the same scores evaluated with 2D imaging in 48.5% and 52.4% of the cases. Similar results were obtained for nephrometry categories (29.7% and 30.7% for PADUA risk and RENAL complexity categories, respectively). The 3D nephrometry score/category demonstrated better accuracy than the 2D nephrometry score/category in predicting overall and major postoperative complications (differences in areas under the curve for each nephrometry score/category were statistically significant comparing the 3D VMs with 2D imaging assessment). Multivariable analyses confirmed 3D PADUA/RENAL nephrometry category as the only independent predictors of overall (P = 0.007; P = 0.003) and major postoperative complications (P = 0.03; P = 0.003).
Conclusions
In the present study, we showed that 3D VMs were more precise than 2D standard imaging in evaluating the surgical complexity of renal masses according to nephrometry score/category. This was attributable to a better perception of tumour depth and its relationships with intrarenal structures using the 3D VM, as confirmed by the higher accuracy of the 3D VM in predicting postoperative complications.
Editorial: Will three‐dimensional models change the way nephrometric scoring is carried out?
There has been an increase in the extent to which imaging is used for preoperative planning of complex urological procedures. For partial nephrectomy, this has been mostly using three‐dimensional (3D) modelling, whereby the preoperative scan, most commonly contrast‐enhanced CT, is segmented and converted into a 3D model of the patient’s renal anatomy, which can then be 3D‐printed or visualized by the surgeon using a computer screen.
In this issue of BJUI, Porpiglia et al. [1] propose the use of 3D models, visualized using a computer for preoperative nephrometric scoring (PADUA and RENAL) of 101 patients to predict postoperative complications. In this preliminary study, they compare the visual scores obtained by two urologists when evaluating only a 3D model, against the scores of two urologists obtained when evaluating only CT images. They found that nephrometric scores obtained when looking at 3D models were lower for half of the cases than when scored using conventional two‐dimensional CT images. Furthermore, they show that for the 101 patients the scores obtained using 3D information were able to give an improved prediction of postoperative complications. The reason for the improved prediction of postoperative complications using 3D modelling is attributed to a better perception of tumour depth and its relationships with intrarenal structures. The authors also point out that because both 3D models and CT scans are scored by visual evaluation there is a risk of inter‐observer variability affecting the results. Overall, this paper introduces an exciting new topic of research in using advanced image analysis techniques for nephrometric scoring.
Many further opportunities exist for developing these ideas of using quantitative image analysis to improve planning and scoring for partial nephrectomy. Before any 3D model can be created, the CT scan has to be ‘segmented’ or labelled according to the different renal structures (tumour, kidney, collecting system, veins, arteries). Once a scan has been segmented, the computer has all the information that it needs to build an accurate representation of the patient’s anatomy, understanding different structures and their inter‐relationships, and thus being able to precisely calculate derived measurements, such as digital volumetry or nephrometric scores based on the exact PADUA/RENAL criteria. Furthermore, novel and more complex nephrometric scores that use segmentation map descriptors could be developed and fitted to postoperative data to further improve predictions. Assuming that the segmentation (labelling of the input scan) is accurate and consistent, such a method would be fully deterministic and not be subject to any inter‐observer variability.
Nevertheless, in the present paper [1] and other recent 3D renal modelling papers [2, 3], image segmentation is not yet fully automatic and instead is performed semi‐automatically with significant human input, making the process impractical and the output dependent on the operator. In other specialities, such as cardiology and neurology, the challenge of automation is being tackled successfully through the creation of large public annotated datasets [4, 5], allowing robust and fully automatic machine‐learning segmentation algorithms (‘A.I.’) to be developed [4]. The creation of a multi‐institutional open‐source dataset of annotated renal CT scans would pave the way for increased research and progress towards automatic, reliable and quantitative image analysis tools for kidney cancer. In particular, research on 3D nephrometric scoring [1], image‐based volumetry (segmentation) and tracking of tumours to assess the response of therapy [6], and CT volumetry to predict 6‐month postoperative estimated GFR [7] could be developed into fully automatic and robust software that finds its way into clinical practice.In conclusion, this paper [1] on 3D models for nephrometric scoring outlines another exciting new way in which advanced image analysis techniques might improve nephrometric scoring and the prediction of complications.
by Lorenz Berger and Faiz Mumtaz
References
- , , et al. Three‐dimensional virtual imaging of the renal tumors: a new tool to improve the accuracy of nephrometric scores. BJU Int 2019; 124: 945-54
- , , et al. Interactive virtual 3D models of renal cancer patient anatomies alter partial nephrectomy surgical planning decisions and increase surgeon confidence compared to volume‐rendered images. Int J Comput Assist Radiol Surg 2019; 14: 723
- , , . The use of 3‐dimensional, virtual reality models for surgical planning of robotic partial nephrectomy. Urology 2019; 125: 92– 7
- , , et al. Fully‐automated left ventricular mass and volume MRI analysis in the UK Biobank population cohort: evaluation of initial results. Int J Cardiovasc Imaging 2018; 34: 281
- , , et al. The multimodal brain tumor image segmentation benchmark (BRATS). IEEE Trans Med Imaging 2015; 34: 1993– 2024
- , , . Assessing tumor response and detecting recurrence in metastatic renal cell carcinoma on targeted therapy: importance of size and attenuation on contrast‐enhanced CT. Am J Roentgenol 2010; 194: 157– 65
- , , et al. Validation of 3‐D volumetric based renal function prediction calculator for nephron sparing surgery. Int Urol Nephrol 2017; 49: 615
Video: Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores
Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores
Abstract
Objectives
To apply the standard PADUA and RENAL nephrometry score variables to three‐dimensional (3D) virtual models (VMs) produced from standard bi‐dimensional imaging, thereby creating three‐dimensional (3D)‐based (PADUA and RENAL) nephrometry scores/categories for the reclassification of the surgical complexity of renal masses, and to compare the new 3D nephrometry score/category with the standard 2D‐based nephrometry score/category, in order to evaluate their predictive role for postoperative complications.
Materials and Methods
All patients with localized renal tumours scheduled for minimally invasive partial nephrectomy (PN) between September 2016 and September 2018 underwent 3D and 2D nephrometry score/category assessments preoperatively. After nephrometry score/category evaluation, all the patients underwent surgery. Chi‐squared tests were used to evaluate the individual patients’ grouping on the basis of the imaging tool (3D VMs and 2D imaging) used to assess the nephrometry score/category, while Cohen’s κ coefficient was used to test the concordance between classifications. Receiver‐operating characteristic curves were produced to evaluate the sensitivity and specificity of the 3D nephrometry score/category vs the 2D nephrometry score/category in predicting the occurrence of postoperative complications. A general linear model was used to perform multivariable analyses to identify predictors of overall and major postoperative complications.
Results
A total of 101 patients were included in the study. The evaluation of PADUA and RENAL nephrometry scores via 3D VMs showed a downgrading in comparison with the same scores evaluated with 2D imaging in 48.5% and 52.4% of the cases. Similar results were obtained for nephrometry categories (29.7% and 30.7% for PADUA risk and RENAL complexity categories, respectively). The 3D nephrometry score/category demonstrated better accuracy than the 2D nephrometry score/category in predicting overall and major postoperative complications (differences in areas under the curve for each nephrometry score/category were statistically significant comparing the 3D VMs with 2D imaging assessment). Multivariable analyses confirmed 3D PADUA/RENAL nephrometry category as the only independent predictors of overall (P = 0.007; P = 0.003) and major postoperative complications (P = 0.03; P = 0.003).
Conclusions
In the present study, we showed that 3D VMs were more precise than 2D standard imaging in evaluating the surgical complexity of renal masses according to nephrometry score/category. This was attributable to a better perception of tumour depth and its relationships with intrarenal structures using the 3D VM, as confirmed by the higher accuracy of the 3D VM in predicting postoperative complications.
December 2019 – About the cover
The lead authors of this month’s selected article (Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores) are from the University of Turin, Italy (UNITO). This university was founded in 1404 making it one of the oldest universities in the World. It has been through some turbulent times but more recently can claim three nobel prize winners: Salvador Luria, Renato Dulbecco and Rita Levi-Montalcini.
The cover image shows the city of Turin at sunset. Turin sits mainly on the Po River and it is surrounded by the Western Alps. As the 10th most visited city in Italy it is known for The shroud of Turin, and its football teams (Juventus and Torino). It is also a cultural centre with many theatres, restaurants, art galleries, palaces, parks and churches.
©istock.com/fabio lamanna
Editorial: Translating cost-utility modelling into the real world – the case of focal high-intensity focussed ultrasound and active surveillance
Health economic modelling is always a challenge. The inputs are never quite what we want them to be. The literature that we have at our disposal suffers from the inevitable deficiencies of lack of maturity, ever diminishing relevance, and questionable applicability as practice evolves. The modelling can never quite reflect the nuances and vagaries of clinical practice. However, the process is an important and in some cases (evaluation by the UK’s National Institute of Clinical and Care Excellence) a necessary one. Knowing the cost of achieving a given health status over a defined time frame is an important consideration in the allocation resource in any finite system of care.
The paper by Bénard et al. [1] is most useful in helping us to understand what the issues are and how our decision-making might impact on cost in the context of low-to-moderate risk prostate cancer.
The issue with these types of analyses is the degree to which the inevitable assumptions made by the investigators are consistent with current practice. Below I have tried to identify some of the areas in which the assumptions diverge from current knowledge and ‘know-how’, in order to illustrate just how difficult the task that Bénard et al. [1] have undertaken.
The first relates to the assumption that both strategies can be applied to the same population. They cannot, or perhaps more correctly – should not. For instance, nobody I know would offer a man focal treatment who had well-characterised micro-focal low-volume Gleason 3+3 (or Gleason Grade Group 1) [2]. We know, from what now constitutes a considerable body of level-1 evidence, that there is no benefit to be derived from intervening in disease that confers little, if any, risk of premature death [3]. Today, focal therapy tends to be applied to men with well-characterised, visually localised Gleason Grade Group ≥2, who want to avoid radical whole gland therapy and the genitourinary side-effects associated with them [4].
The second relates to the synergies between the two treatments. Increasingly men who opt for active surveillance (AS) upfront have an increasing tendency to opt for focal treatment on radiological progression of any lesion under scrutiny. This makes quite a bit of intuitive sense. These are men who appear comfortable with the process of observation, are likely to place high utility on genitourinary function, may have exhibited a very stable background prostate (apart from the expanding lesion depicted on MRI), are likely to be very well informed, and will, by now, be very well-characterised histologically. These, as it happens, are the ideal attributes for a candidate for focal therapy.
The third is a reflection on the relevance of the literature to inform the question being posed. It is no fault of the authors that AS has changed beyond recognition in the last few years. This change has been driven by the use of MRI in the risk stratification process for candidate selection, the substation of temporal biopsy assessment by imaging and the reduction, and at times elimination, of the re-classification vs progression error that confounds most of the literature on
surveillance. Modelling events on historical single-institution cohorts (as AS has never been evaluated in a randomised setting apart from one comparison against focal therapy) is probably unhelpful in helping us to understand and inform our future [5].
The fourth concerns scope. Why limit this analysis to focal high-intensity focussed ultrasound? All focal therapies, irrespective of energy source, seem to produce very similar outcomes, both in terms of freedom from failure (time to radical treatment and/or metastasis) and in relation to preservation of genitourinary function. Broadening the scope, by including vascular targeted photo-therapy and cryotherapy, would have meant that randomised trials could have been
included as inputs, with the effect of possibly reducing the high levels of uncertainty that bedevil the current analysis [5,6].
The fifth recognises the dynamic nature of the progression risk in AS cohorts. This is an important, but poorly recognised, attribute of the mature AS cohorts that we tend to rely upon. These cohorts are dynamic entities that have as entrants men of increasingly lower risk (due to a recent improvement in risk stratification) and, at the same time, continually exit the very men with the highest risk, i.e., the ‘progressors’. Thus, over time, the cohort undergoes a gradual, but inevitable, reduction in risk. The more mature the cohort, the greater the reduction. By referencing mature cohorts (when trying to predict the fate of future patients) we
will, therefore, have a tendency to over-estimate the benefit/safety of AS in a contemporary setting.
This is not to say that we should not endeavour to estimate the cost of achieving a given health state. We need this, perhaps more than ever. What we need to strive towards are models that represent both the reality of practice and the very latest, and most subtle, distillation of the current evidence.
by Mark Emberton
References
- Bénard A, Duroux T, Robert G. Cost-utility analysis of focal high-intensity focussed ultrasound vs active surveillance for low- to intermediate-risk prostate cancer using a Markov multi-state model. BJU Int 2019; 124: 962–71
- Klotz L, Emberton M. Management of low risk prostate cancer-active surveillance and focal therapy. Nat Rev Clin Oncol 2014; 11: 324–34
- Hamdy FC, Donovan JL, Lane JA et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med 2016; 375: 1415–24
- Elliott D, Hamdy FC, Leslie TA et al. Overcoming difficulties with equipoise to enable recruitment to a randomised controlled trial of partial ablation vs radical prostatectomy for unilateral localised prostate cancer. JU Int 2018; 122: 970–7
- Azzouzi AR, Vincendeau S, Barret E et al. Padeliporfin vascular-targeted photodynamic therapy versus active surveillance in men with low-risk prostate cancer (CLIN1001 PCM301): an open-label, phase 3, randomised controlled trial. Lancet Oncol 2017; 18: 181–91
- Donnelly BJ, Saliken JC, Brasher PM et al. A randomized trial of external beam radiotherapy versus cryoablation in patients with localized prostate cancer. Cancer 2010; 116: 323–30
Residents’ podcast: NICE Guidance – Transurethral water jet ablation for lower urinary tract symptoms caused by benign prostatic hyperplasia
Nikita Bhatt is a Specialist Trainee in Urology in the East of England Deanery and a BURST Committee member @BURSTUrology
NICE Guidance – Transurethral water jet ablation for lower urinary tract symptoms caused by benign prostatic hyperplasia
Recommendations
- 1.1 The evidence on transurethral water jet ablation for lower urinary tract symptoms (LUTS) caused by benign prostatic hyperplasia (BPH) raises no major safety concerns. The evidence on efficacy is limited in quantity. Therefore, this procedure should only be used with special arrangements for clinical governance, consent, and audit or research.
- 1.2 Clinicians wishing to do transurethral water jet ablation for LUTS caused by BPH should:
- Inform the clinical governance leads in their NHS trusts.
- Ensure that patients understand the uncertainty about the procedure’s efficacy and provide them with clear written information to support shared decision‐making. In addition, the use of the National Institute for Health and Care Excellence (NICE) information for the public is recommended.
- Audit and review clinical outcomes of all patients having transurethral water jet ablation for LUTS caused by BPH. NICE has identified relevant audit criteria and has developed an audit tool (which is for use at local discretion).
- 1.3 The procedure should only be done by clinicians who have been trained in the technique.
- 1.4 NICE encourages further research into transurethral water jet ablation for LUTS caused by BPH and may update the guidance on publication of further evidence. Further research should report long‐term follow‐up and include re‐intervention rates.
BJUI Podcasts now available on iTunes, subscribe here https://itunes.apple.com/gb/podcast/bju-international/id1309570262
Article of the week: Cost–utility analysis of focal-HIFU vs AS for low‐ to intermediate‐risk PCa using a Markov multi‐state model
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urology community and a video prepared by the authors; we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Cost–utility analysis of focal high‐intensity focussed ultrasound vs active surveillance for low‐ to intermediate‐risk prostate cancer using a Markov multi‐state model
Antoine Bénard*†, Thomas Duroux* and Gregoire Robert‡
*Univ. Bordeaux, Inserm, UMR 1219, Bordeaux Population Health Research Center, Team EMOS, †CHU de Bordeaux, Pôle de santé publique, Service d’information Médicale, USMR & CIC-EC 14-01, and ‡CHU de Bordeaux, Service d’urologie, Andrologie et Transplantation Renale, Université de Bordeaux, Bordeaux, France
Abstract
Objectives
To estimate the relative cost‐effectiveness of focal high‐intensity focussed ultrasound (F‐HIFU) compared to active surveillance (AS) in patients with low‐ to intermediate‐risk prostate cancer, in France.
Patients and Methods
A Markov multi‐state model was elaborated for this purpose. Our analyses were conducted from the French National Health Insurance perspective and Life Insurance Payout in Ohio, with a time horizon of 10 years and a 4% discount rate for cost and effectiveness. A secondary analysis used a 30‐year time horizon. Costs are presented in 2016 Euros (€), and effectiveness is expressed as quality‐adjusted life years (QALYs). Model parameters’ value (probabilities for transitions between health states, and cost and utility of health states) is supported by systematic literature reviews (PubMed) and random effect meta‐analyses. The cost of F‐HIFU in our model was the temporary tariff attributed by the French Ministry of Health to the overall treatment of prostate cancer by HIFU (€6047).
Our model was analysed using Microsoft Excel 2010 (Microsoft Corp., Redmond, WA, USA). Uncertainty about the value of the model parameters was handled through probabilistic analyses.
Results
The five health states of our model were as follows: initial state (AS or F‐HIFU), radical prostatectomy, radiation therapy, metastasis, and death.
Transition probabilities from the initial F‐HIFU state relied on four articles eligible for our meta‐analyses. All were non‐comparative studies. Utilities relied on a single cohort in San Diego, CA, USA.
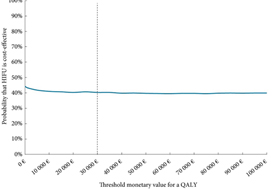
For a fictive cohort of 1000 individuals followed for 10 years, F‐HIFU would be €207 520 more costly and would yield 382 less QALYs than AS, which means that AS is cost‐effective when compared to F‐HIFU. For a threshold value varying from €0 to 100 000/QALY, the probability of AS being cost‐effective compared to F‐HIFU varied from 56.5% to 60%. This level of uncertainty was in the same range with a 30‐year time horizon.
Conclusion
Given existing published data, our results suggest that AS is cost‐effective compared to F‐HIFU in patients with low‐ and intermediate‐risk prostate cancer, but with high uncertainty. This uncertainty must be scaled down by continuing to supply the model with new published data and ideally through a randomised clinical trial that includes cost‐effectiveness analyses.
Residents’ podcast: NICE Guidance – Prostate cancer: diagnosis and management
Mr Joseph Norris is a Specialty Registrar in Urology in the London Deanery. He is currently undertaking an MRC Doctoral Fellowship at UCL, under the supervision of Professor Mark Emberton. His research interest is prostate cancer that is inconspicuous on mpMRI. Joseph sits on the committee of the BURST Research Collaborative as the Treasurer and BSoT Representative.
NICE Guidance – Prostate cancer: diagnosis and management
Context
Prostate cancer is the most common cancer in men, and the second most common cancer in the UK. In 2014, there were over 46,000 new diagnoses of prostate cancer, which accounts for 13% of all new cancers diagnosed. About 1 in 8 men will get prostate cancer at some point in their life. Prostate cancer can also affect transgender women, as the prostate is usually conserved after gender-confirming surgery, but it is not clear how common it is in this population.
More than 50% of prostate cancer diagnoses in the UK each year are in men aged 70 years and over (2012), and the incidence rate is highest in men aged 90 years and over (2012 to 2014). Out of every 10 prostate cancer cases, 4 are only diagnosed at a late stage in England (2014) and Northern Ireland (2010 to 2014). Incidence rates are projected to rise by 12% between 2014 and 2035 in the UK to 233 cases per 100,000 in 2035.
A total of 84% of men aged 60 to 69 years at diagnosis in 2010/2011 are predicted to survive for 10 or more years after diagnosis. When diagnosed at the earliest stage, virtually all people with prostate cancer survive 5 years or more: this is compared with less than a third of people surviving 5 years or more when diagnosed at the latest stage.
There were approximately 11,000 deaths from prostate cancer in 2014. Mortality rates from prostate cancer are highest in men aged 90 years and over (2012 to 2014). Over the past decade, mortality rates have decreased by more than 13% in the UK. Mortality rates are projected to fall by 16% between 2014 and 2035 to 48 deaths per 100,000 men in 2035.
People of African family origin are at higher risk of prostate cancer (lifetime risk of approximately 1 in 4). Prostate cancer is inversely associated with deprivation, with a higher incidence of cases found in more affluent areas of the UK.
Costs for the inpatient treatment of prostate cancer are predicted to rise to £320.6 million per year in 2020 (from
£276.9 million per year in 2010).
This guidance was updated in 2014 to include several treatments that have been licensed for the management of
hormone-relapsed metastatic prostate cancer since the publication of the original NICE guideline in 2008.
Since the last update in 2014, there have been changes in the way that prostate cancer is diagnosed and treated. Advances in imaging technology, especially multiparametric MRI, have led to changes in practice, and new evidence about some prostate cancer treatments means that some recommendations needed to be updated.
BJUI Podcasts are available on iTunes, subscribe here https://itunes.apple.com/gb/podcast/bju-international/id1309570262