An unusual case of granulomatous prostatitis with caseification necrosis: Primary Tuberculous Prostatitis in a young male
Primary prostatic tuberculosis is a very rare form of the tuberculosis infection. Isolated prostate tuberculosis is generally seen in immunocompromised patients and in those of middle or advanced age. We report a case of isolated prostatic tuberculosis in a young, healthy and immunocompetent patient.
Authors:
Inci, Mehmet; Yula, Erkan; Inci, Melek; Rifaioglu, Murat; Ozgur, Tumay; Davran, Ramazan; Erden, Ersin
Corresponding Author: Mehmet Inci. Mustafa Kemal University Faculty of Medicine – Department of Urology, Hatay, Turkey
INTRODUCTION
The genitourinary system is the most frequently affected region of the body in extra-pulmonary tuberculosis. The kidneys, ureters, bladder or genital organs are usually involved [1] Tuberculosis of the prostate has mainly been described in immunocompromised patients [2-4]. Tuberculosis is rare in the prostate and it is coincidentally diagnosed during pathological evaluation after transurethral resection in many cases [5]. The most commonly involved pathogen is Mycobacterium tuberculosis. It is thought that involvement of the prostate with tuberculosis usually results from hematogenous spread, although it may occur due to organisms descending from kidneys or local spread from the genital tract [1, 6].
CASE
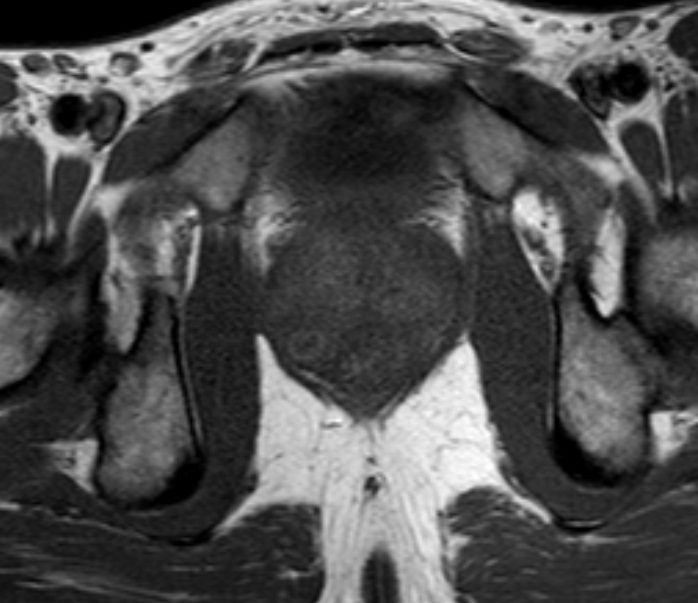
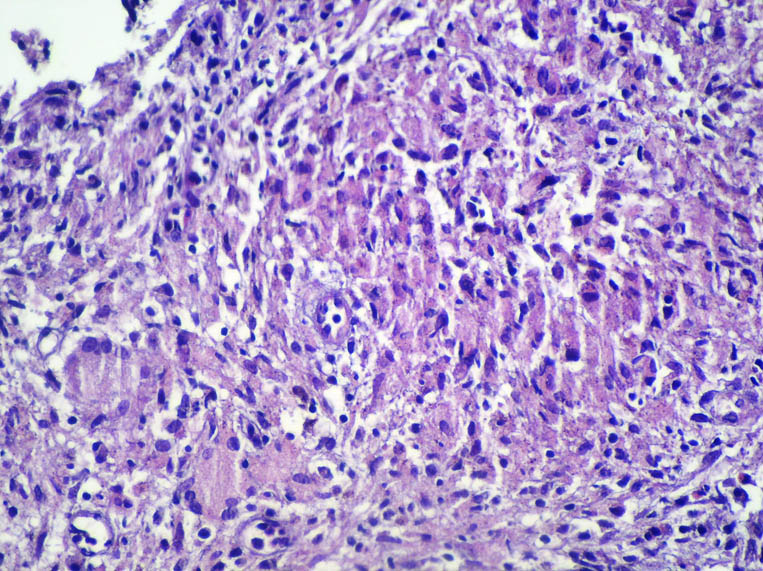
A 22-year old man who had been diagnosed as having acute prostatitis and received quinolone for one month without improvement in another facility was referred to the Medical Center of Mustafa Kemal University, Medicine School with features of prostatism and lower urinary tract symptoms. On digital rectal examination (DRE), the prostate was found to be enlarged, hard, irregular and nodular. It was tender to palpation. The patient was not known to have any other medical problems. There were no abnormalities on physical examination. Blood tests revealed a white cell count of 6200/mm3, hematocrit of 43% and an erythrocyte sedimentation rate of 63 mm/h. The patient’s serum total prostate-specific antigen (tPSA) level was elevated and found to be 7.867 ng/ml. On urinalysis, there was no finding other than pyuria. Direct urinary system radiography was normal. On the magnetic resonance imaging (MRI), there was low signal intensity at the peripheral zone of prostate in T1-weighted sequences, (Figure 1). Transrectal ultrasound (TRUS) examination was performed with a Sonoline Elegra Scanner (Siemens Ultrasound Division, Mountain View, CA) equipped with a 6.5-MHz endorectal probe. TRUS showed a markedly enlarged prostate, with a heterogeneous and hypoechoic appearance to the parenchyma and enlargement of the seminal vesicles. TRUS guided systematic biopsies of the prostate were performed after sonographic examination. The specimens were diagnosed histopathologically as granulomatous prostatitis, with granuloma consisting of areas of focal caseating necrosis, Langhans giant cells and epitheloid histocytes (Figure 2). We also performed intravenous urography. There were no pathological findings seen in the relevant structures of the urinary tract. Both HIV-antibody tests and the tuberculin skin test were negative. No pathological findings were seen on chest X-ray and CT of the thorax. Urine samples were taken over consecutive 3 days. Ehrlich-Ziehl-Neelsen (EZN) staining and culture was performed on these samples. No acid fast bacilli were observed on EZN staining. No growth was detected after incubation on Middlebrook agar (BacT/Alert 3D system; BacT/ALERT MP Bottle supplemented with Middlebrook 7H9 Broth) and Lowenstein-Jensen medium for eight weeks. In addition; prostate secretions were collected by massage. No microorganisms were seen in this sample on Gram staining, while 5-7 leukocytes were seen in each field (x40 magnification) on Giemsa staining. In addition, acid fast bacilli were seen in EZN staining, but no growth was detected on culture of the prostate secretion on Middlebrook agar and Lowenstein-Jensen medium for 8 weeks. Diagnosis of prostatic tuberculosis was made based on the pathology results and acid fast bacilli positivity. The patient was treated with isoniazid, rifampicin, pyrazimanide and ethambutol for the initial two months; and he received isoniazid and rifampicin for a further four months. His clinical signs improved after treatment. Control MR imaging showed that prostate returned to its normal size after therapy and his tPSA level reduced to 0.8 ng/ml.
Figure 1
Figure 2
DISCUSSION
Tuberculosis prostatitis is an uncommon benign inflammatory disease of the prostate that rarely presents clinically in urological practice. Recognizing this condition is important, due to the possibility of it being misdiagnosed as prostatic carcinoma, both on clinical examination and TRUS [7, 8]. The diagnosis of tuberculosis prostatitis can be made via histopathologic and microbiological examination.
Tuberculous prostatitis has a specific histopathologic appearance; the initial lesion begins in stroma, where it spreads to the acini in most cases. In mature lesions, there is usually focal caseation with an incomplete fibrous capsule, epitheloid cells and lymphocytes. Multi-nucleated giant cells may also be observed. In our case, there were granulomas consisting of focal caseating necrosis areas, Langhans giant cells and epitheloid histiocytes, suggesting prostatic tuberculosis.
In tuberculosis of the prostate, acid fast bacilli screening and culture of prostatic secretions are important for diagnosis. The sensitivity of serial urine and semen cultures is 50%. The rate of bacillus identification was reported as 52% in samples of prostate secretions [9]. Although acid fast bacilli were observed in the EZN staining of prostate secretion in our case, no growth was detected in culture. We believe that this could be explained by previous quinolone use [10].
Sterile pyuria is a classic feature of genitourinary tuberculosis. In our caseon urinalysis, there was pyuria and no growth was detected on urine culture.
It has been reported that a tuberculosis focus is founded in any area of the genitourinary system in 75-90% of the cases with prostate involvement, while prostate involvement is detected in 3-12% of the cases with systemic tuberculosis [5]. Imaging studies may be helpful to identify the presence of concurrent tuberculosis of other organs. Thus TRUS, intravenous urography (IVU), MRI and chest X-ray should be considered in such patients. TRUS can demonstrate enlarged gland with solitary or multiple hypoechoic areas of varying size [2, 7, 8]. MR imaging may be helpful in the differential diagnosis and some characteristic of prostatic tuberculosis have been established [1, 3]. There may be lesions with low signal intensity on MRI, suggesting prostatic tuberculosis. In our case, similar findings were observed on both MRI and TRUS in accordance with the above-mentioned studies. Although there are abnormal findings on IVU in most cases of genitourinary tuberculosis [11], there was no finding suggestive of urinary tuberculosis of the kidneys or bladder on IVU in our case.
Extra-pulmonary involvement is seen in 50-70% of immunocompromised patients, particularly in those with HIV infection, isolated prostatic tuberculosis is uncommon in patients with immune competency [2-4]. Also in our patient anti-HIV testing was negative and his immune system was normal
Primary tuberculosis of the prostate has mainly been described in middle or advanced age, (46- 65 old years) [12 -15]. On the other hand in our case was in a man 22-years of age.
The clinical findings of prostatic tuberculosis are often nonspecific, and the most common symptoms are of lower urinary tract obstruction. In patients with tuberculosis prostatitis, the prostate gland is firm, irregular, nodular or granular on digital rectal examination [3,7,8,16 ]. Our patient had similar findings on clinical examination. Tuberculosis prostatitis may cause elevation of PSA levels [7, 8, and 17], thus mimicking prostatic carcinoma. However, studies have shown that this rise in PSA levels is transient and decreases with resolution of inflammation [18]. The patient’s PSA level was elevated and decreased into normal limits after anti-tuberculosis therapy.
In our case, the upper urinary tract and bladder were normal on IVP and MR imaging. There was no evidence of tuberculosis at pulmonary or extra-pulmonary sites. The diagnosis of primary prostatic tuberculosis was made based on acid fast bacilli positivity in prostate secretiosn and on pathological findings.
In conclusion, although isolated prostatic tuberculosis is generally seen in immunocompromised patients and in those of middle or advanced age, it should be kept in mind that it can be present in young adults with immune competency refractory to routine therapies. We suggest that TRUS-guided prostate biopsy is an important tool for the diagnosis of prostatic tuberculosis and is essential to distinguish it from carcinoma.
References
1. Lenk S, Schroeder J. Genitourinary tuberculosis. Curr Opin Urol. 2001; 11: 93-8.
2. Gebo KA. Prostatic tuberculosis in an HIV infected male. Sex Transm Infect. 2002; 78: 147-8.
3. Trauzzi SJ, Kay CJ, Kaufman DG, et al. Management of prostatic abscess in patients with human immunodeficiency syndrome. Urology 1994; 43: 629.
4. Fujikawa K, Matsui Y, Fukuzawa S, et al. A case of tuberculosis of the prostate. Scand J Urol Nephrol 1999; 33: 268.
5. Gow JG: Genitourinary tuberculosis. In: Walsh PC, Retik AB, Stamey TA, Vaughan ED, eds. Campbell’s Urology. 7th ed. Philadelphia, Pa: WB Saunders; 1998: 807-36.
6. Gorsc GJ, Belshe RB. Male genital tuberculosis: a review of the literature with instructive case reports. Rev Infect Dis. 1985; 7: 511.
7. Clements R, Gower TK, Griffiths GJ, et al. Transrectal ultrasound appearances of granulomatous prostatitis. Clin Radiol 1993; 47: 174.
8. Naik KS, Carey BM. The transrectal ultrasound and MRI appearances of granulomatous prostatitis and its differentiation from carcinoma. Clin Radiol. 1999; 54: 173.
9. Tanagho EA. Specific infections of the genitourinary tract. In: Tanagho EA, McAnich JW, eds. Smith’s General Urology. 15th ed. New York, NY: McGraw-Hill Professional Publishing, 2000: 265-81.
10. Vachera S, Pellegrina JL, Leblancb F, Fourchea J, Maugeina J. Comparative antimycobacterial activities of ofloxacin, ciprofloxacin and grepafloxacin. J Antimicrob Chemother. 1999; 44: 647-52.
11. Wise GJ, Marella VK. Genitourinary manifestations of tuberculosis. Urol Clin North Am. 2003; 30: 111-21.
12. Tazi K, Nouri M, Elkhadir K, el Ghorfi A, Ibnattya A, Hachimi M, Lakrissa A. Prostatic tuberculosis. Report of 2 cases. Ann Urol (Paris). 1999; 33: 274-6.
13. López Barón E, Gómez-Arbeláez D, Díaz-Pérez JA. Primary prostatic tuberculosis. Case report and bibliographic review. Arch Esp Urol. 2009; 62: 309-13.
14. Rabesalama SS, Rakoto-Ratsimba HN, Rakototiana AF, Razafimahatratra R, Raherison RA, Rantomalala HY, Randrianjafisamindrakotroka NS. Isolated prostate tuberculosis. Report of a case in Madagascar. Prog Urol. 2010; 20: 314-6.
15. Tamsel S, Killi R, Ertan Y, Demirpolat G. A rare case of granulomatous prostatitis caused by Mycobacterium tuberculosis. J Clin Ultrasound. 2007; 35: 58-61.
16. Mohan H, Bal A, Punia RPS, et al. Granulomatous prostatitis: an infrequent diagnosis. Int J Urol. 2005; 12: 474.
17. Liu S, Miller PD, Holmes SA, et al. Eosinophilic prostatitis and prostate specific antigen. Br J Urol. 1992; 69: 61.
18. Speights VO, Brawn PN. Serum prostate specific antigen levels in non-specific granulomatous prostatitis. Br J Urol. 1996; 77: 408.
Date added to bjui.org: 18/04/2013
DOI: 10.1002/BJUIw-2012-052-web