Article of the Week: Sitting time, physical activity and the risk of LUTS
Every Week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Sitting time, physical activity and the risk of lower urinary tract symptoms: a cohort study
Abstract
Objectives
To examine the association of sitting time and physical activity level with the incidence of lower urinary tract symptoms (LUTS) in a large sample of Korean men.
Materials and Methods
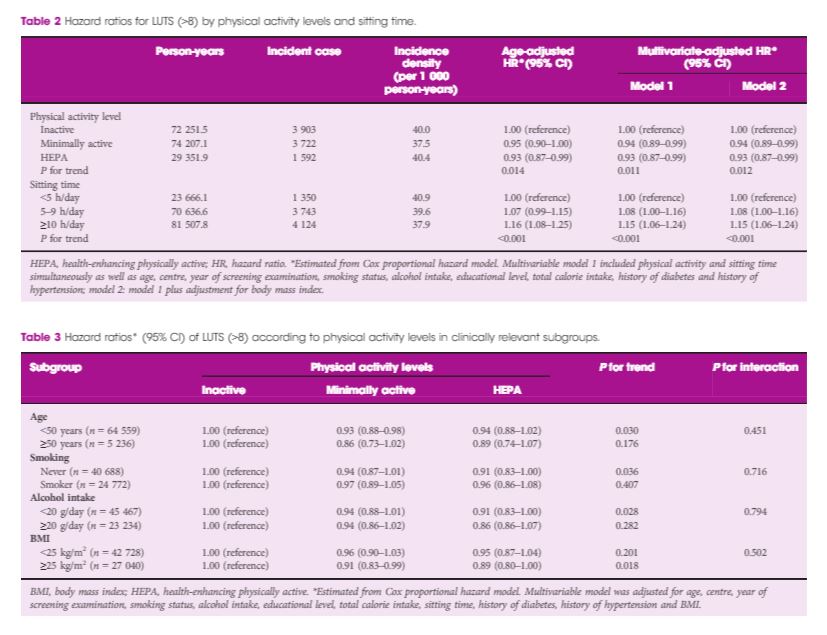
A cohort study was performed in 69 795 Korean men, free of LUTS at baseline, who were followed up annually or biennially for a mean of 2.6 years. Physical activity level and sitting time were assessed using the validated Korean version of the International Physical Activity Questionnaire Short Form. LUTS were assessed using the International Prostate Symptom Score (IPSS) and clinically significant LUTS were defined as an IPSS score ≥8.
Results
Over 175 810.4 person‐years, 9 217 people developed significant LUTS (incidence rate, 39.0 per 1 000 person‐years). In a multivariable‐adjusted model, both low physical activity level and prolonged sitting time were independently associated with the incidence of LUTS. The hazard ratios (95% confidence intervals [CIs]) for incident LUTS comparing minimally active and health‐enhancing physically active groups vs the inactive group were 0.94 (95% CI 0.89–0.99) and 0.93 (95% CI 0.87–0.99), respectively (P for trend 0.011). The hazard ratios (95% CIs) for LUTS comparing 5–9 and ≥10 h/day sitting time vs <5 h/day were 1.08 (95% CI 1.00–1.24) and 1.15 (95% CI 1.06–1.24), respectively (P for trend <0.001).
Conclusions
Prolonged sitting time and low physical activity levels were positively associated with the development of LUTS in a large sample of middle‐aged Korean men. This result supports the importance of both reducing sitting time and promoting physical activity for preventing LUTS.