Article of the Week: Significance of LVI in organ-confined, node-negative UCB – the p53-MVAC trial
Every Week the Editor-in-Chief selects the Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Significance of lymphovascular invasion in organ-confined, node-negative urothelial cancer of the bladder: data from the prospective p53-MVAC trial
OBJECTIVES
To investigate the association between lymphovascular invasion (LVI) and clinical outcome in organ-confined, node-negative urothelial cancer of the bladder (UCB) in a post hoc analysis of a prospective clinical trial. To explore the effect of adjuvant chemotherapy with methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) on outcome in the subset of patients whose tumours exhibited LVI.
PATIENTS AND METHODS
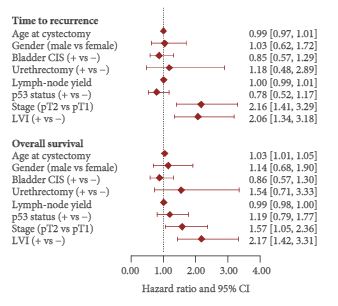
Surgical and tumour factors were extracted from the operative and pathology reports of 499 patients who had undergone radical cystectomy (RC) for pT1–T2 N0 UCB in the p53-MVAC trial (Southwest Oncology Group 4B951/NCT00005047). The presence or absence of LVI was determined by pathological examination of transurethral resection or RC specimens. Variables were examined in univariate and multivariate Cox proportional hazards models for associations with time to recurrence (TTR) and overall survival (OS).
RESULTS
Among 499 patients with a median follow-up of 4.9 years, a subset of 102 (20%) had LVI-positive tumours. Of these, 34 patients had pT1 and 68 had pT2 disease. LVI was significantly associated with TTR with a hazard ratio (HR) of 1.78 [95% confidence interval (CI) 1.15–2.77; number of events (EV) 95; P = 0.01) and with OS with a HR of 2.02 (95% CI 1.31–3.11; EV 98; P = 0.001) after adjustment for pathological stage. Among 27 patients with LVI-positive tumours who were randomised to receive adjuvant chemotherapy, receiving MVAC was not significantly associated with TTR (HR 0.70, 95% CI 0.16–3.17; EV 7; P = 0.65) or with OS (HR 0.45, 95% CI 0.11–1.83; EV 9; P = 0.26).
CONCLUSIONS
Our post hoc analysis of the p53-MVAC trial revealed an association between LVI and shorter TTR and OS in patients with pT1–T2N0 disease. The analysis did not show a statistically significant benefit of adjuvant MVAC chemotherapy in patients with LVI, although a possible benefit was not excluded.