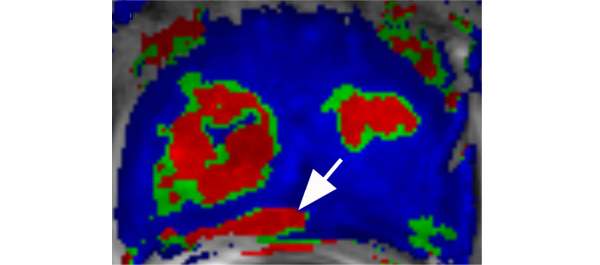
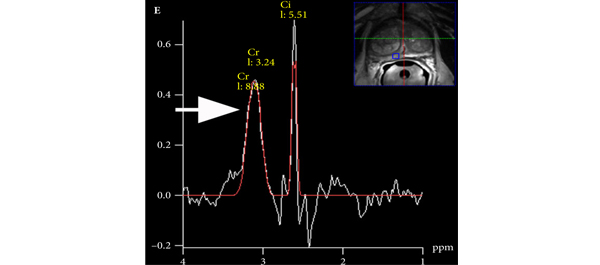
Editorial: Multiparametric MRI and active surveillance for prostate cancer: future directions
A growing body of data exists suggesting an important role of MRI in selecting men with prostate cancer for active surveillance (AS). In the present study, Park et al. [1] show that a suspicious lesion on MRI was independently predictive of adverse pathology after radical prostatectomy (RP). This finding supports existing data suggesting that suspicious lesions on MRI confer an increased risk of disease reclassification among men enrolled in AS [2]. Indeed, in our institutional AS experience we found that men with a suspicious lesion on MRI were more likely to have biopsy reclassification with extended follow-up [3].While these data are provocative, much work remains to be done before the adoption of MRI as a standard screening tool for entry into AS for men with very low-risk prostate cancer.
Introduction of functional sequence imaging into multiparametric MRI protocols has resulted in improved detection and characterisation of clinically localised prostate cancer. However, before widespread implementation into AS protocols can occur, increased rigor and standardisation in image interpretation is needed. As in the present study, 5-point Likert scales have become an increasingly popular method of quantifying a lesions likelihood of representing cancer [1]. Still other authors have quantified a lesions level of suspicion using both weighted and non-weighted scoring systems based on the number of positive MRI sequences [3,4]. While useful for statistical analysis, these reporting methods are fraught with concerns of inter-observer variability and generalizability. Additionally, a recent report by Lee et al. [5] found that a simple measurement of lesion diameter on diffusion-weighted MRI was predictive of insignificant disease after RP. Combining the plethora of functional and morphological data obtained by multiparametric MRI into a standardised, reproducible tool will greatly facilitate implementation of MRI into current AS screening protocols.
As a step in the right direction, Stamatakis et al. [4] recently generated a nomogram for predicting biopsy reclassification in men on AS after taking into consideration both functional and morphological characteristics of MRI lesions. Adding an additional layer of complexity, they also assessed the utility of calculated values, e.g. lesion density (lesion volume/prostate volume), in predicting biopsy reclassification. Briefly, their analysis showed that the number of lesions, lesion suspicion, and lesion density were predictive of biopsy reclassification. While nomogram validation and testing of its predictive value on pathological outcomes is needed, this represents a major advance in the standardised application of MRI to AS cohorts.
Despite great strides in the application of multiparametric MRI to AS cohorts, a significant concern about the false-negative rate exists. Considering the present report, of the 35 men with no visible lesion on MRI, 14.3% men had unfavourable pathology after RP [1]. This is similar to previous studies reporting disease reclassification rates of <18% [2,6]. These men with normal imaging and high-grade cancer highlight the importance of incorporating imaging and clinical data when selecting men for AS. Better defining the false-negative rate of multiparametric MRI, and effectively identifying men with a normal MRI and high-grade disease remain major challenges.
Considering all of the available data, it is becoming increasingly clear that MRI has the potential for improving the identification of patients for whom AS would be safe. It is currently the practice at our institution to refer eligible men for multiparametric MRI before enrolment in AS. Our future scholarly efforts should be directed at the standardisation of reporting MRI data and the development of user-friendly AS criteria that synthesise MRI results with clinicopathological data.
Jeffrey K. Mullins and H. Ballentine Carter
James Buchanan Brady Urological Institute, Johns Hopkins Medical Institutions, Baltimore, MD, USA
References
- Park BH, Jeon HG, Choo SH et al. Role of multiparametric 3.0-Tesla magnetic resonance imaging in patients with prostate cancer eligible for active surveillance. BJU Int 2014; 113: 864–70
- Margel D, Yap SA, Lawrentschuk N et al. Impact of multiparametric endorectal coil prostate magnetic resonance imaging on disease reclassification among active surveillance candidates: a prospective cohort study. J Urol 2012; 187: 1247–52
- Mullins JK, Bonekamp D, Landis P et al. Multiparametric magnetic resonance imaging findings in men with low-risk prostate cancer followed using active surveillance. BJU Int 2013; 111: 1037–45
- Stamatakis L, Siddiqui MM, Nix JW et al. Accuracy of multiparametric magnetic resonance imaging in confirming eligibility for active surveillance for men with prostate cancer. Cancer 2013; 119: 3359–66
- Lee DH, Koo KC, Lee SH et al. Tumor lesion diameter on diffusion weighted magnetic resonance imaging could help predict insignificant prostate cancer in patients eligible for active surveillance: preliminary analysis. J Urol 2013; 190: 1213–7
- Guzzo TJ, Resnick MJ, Canter DJ et al. Endorectal T2-weighted MRI does not differentiate between favorable and adverse pathologic features in men with prostate cancer who would qualify for active surveillance. Urol Oncol 2012; 30: 301–5