Editorial: Do all patients with renal cell carcinoma need a chest computed tomography?
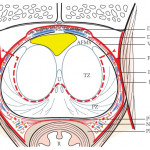
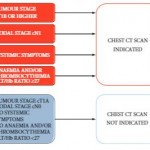
While all patients with RCC need chest imaging for staging evaluation, the answer to the question in the title is ‘No’, and, in fact, many patients would be adequately staged with a chest X-ray, albeit with reduced accuracy. Evidence to support this assertion is provided by Larcher et al. [1] in this issue of BJUI, who retrospectively evaluated 1946 patients with a solitary and sporadic RCC mass. While excluding patients who did not have surgery and those with visceral metastases seen on abdominal imaging, the authors observed pulmonary metastases in 6% (119 patients) of their population. In a multivariable analysis, features associated with a positive chest CT included cT1b+, cN1, systemic symptoms, anaemia, and thrombocytosis. Incorporating these features into a predictive model, the authors report a robust concordance index of 0.88, with the effect of each feature demonstrated in a nomogram. Further, the authors report that if a chest CT is only performed when the risk of a positive result is >1%, 37% of their population could have been spared a chest CT while missing a positive result in only 0.2% (four patients). Patient factors that predict for a <1% risk of a positive chest CT essentially include those with cT1aN0 RCC without systemic symptoms, anaemia, or thrombocytosis. Thus, the authors conclude that in these low-risk patients, a chest CT can be omitted, while any patient that is cT1b+, cN1, or with systemic symptoms, anaemia, or thrombocytosis warrants a dedicated chest CT at diagnosis.
The finding that patients with RCC with smaller tumours (cT1a or ≤4 cm) were unlikely to harbour pulmonary metastases is consistent with prior literature. Observations from the Memorial Sloan-Kettering Cancer Center (MSKCC) [2], and subsequently validated by our group at Mayo Clinic [3], suggested that among surgically treated patients with RCC, risk of M1 disease (at any location) at diagnosis was non-existent for tumours of <2 cm, was <1% for tumours of 2–3 cm, and was only 1–2% for tumours of 3-4 cm in size. Given that it is rare for patients with small renal masses to endorse systemic symptoms or have paraneoplastic symptoms related to the tumour, these prior observations suggest a lack of utility for chest CT for patients with small renal masses supporting the findings from Larcher et al. [1].
In patients with RCC with synchronous metastases, lung is the most common site of spread and guidelines uniformly recommend chest imaging at diagnosis. However, a ‘select-all’ strategy for chest CT in patients with renal masses leads to unnecessary findings in those with a benign primary tumour, increased use of healthcare resources, and relatively frequent findings of indeterminate lesions. In fact, contemporary observations from the MSKCC found that about half of patients with RCC undergoing surgery had indeterminate pulmonary nodules on chest CT that required either additional evaluation or subsequent chest CT to document stability [4]. Further, the presence of indeterminate pulmonary nodules was not associated with distant metastases or death from RCC after surgery unless they were >1 cm, which only represented a small portion (4%) of the entire cohort [4]. Thus, the analysis from Larcher et al. [1] in this issue of BJUI has meaningful clinical relevance; that is, patients with cT1aN0 RCC without symptoms or laboratory abnormalities do not require a chest CT for screening of their lungs.