Article of the week: Pneumonitis should not prevent continued mTOR inhibitor use
Every week the Editor-in-Chief selects the Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by prominent members of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Mammalian target of rapamycin (mTOR) inhibitor-associated non-infectious pneumonitis in patients with renal cell cancer: predictors, management, and outcomes
Bradley J. Atkinson, Diana H. Cauley, Chaan Ng*, Randall E. Millikan†, Lianchun Xiao‡, Paul Corn†, Eric Jonasch† and Nizar M. Tannir†
Departments of Pharmacy Clinical Programs, *Diagnostic Radiology, †Genitourinary Medical Oncology and ‡Biostatistics, University of Texas MD Anderson Cancer Center, Houston, TX, USA
B.J.A. and D.H.C. are co-first authors
Supported in part by a Cancer Center Support Grant (CA016672) from the National Institutes of Health.
OBJECTIVE
• To characterise the incidence, onset, management, predictors, and clinical impact of mammalian target of rapamycin (mTOR) inhibitor-associated non-infectious pneumonitis (NIP) on patients with metastatic renal cell carcinoma (mRCC).
PATIENTS AND METHODS
• Retrospective review of 310 patients with mRCC who received temsirolimus and/or everolimus between June 2007 and October 2010.
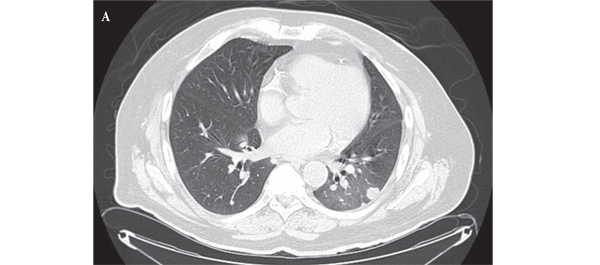
• Clinical correlations were made with serial radiological imaging.
• Fisher’s exact, Wilcoxon rank-sum, and logistic regression analyses were used to evaluate the association of NIP with demographic or clinical factors.
• Log-rank and Cox proportional hazards regression analyses were used for the time-to-event analysis.
RESULTS
• NIP occurred in 6% of temsirolimus-treated and 23% of everolimus-treated patients. Symptoms included cough, dyspnoea, and fever (median of two and three symptoms per patient, respectively).
• The median National Cancer Institute Common Toxicity Criteria for Adverse Events pneumonitis grade was 2 for both groups.
• Older age and everolimus treatment were predictive of NIP.
• Patients who developed NIP had a significantly longer time on treatment (median 4.1 vs 2 months) and overall survival (OS) (median 15.4 vs 7.4 months).
• NIP was a predictor of improved OS by multivariate analysis.
CONCLUSIONS
• There was an increased incidence of NIP in everolimus-treated patients.
• Improved OS in patients who developed NIP is an intriguing finding and should be further investigated. Given the incidence, morbidity, and outcomes seen in patients on everolimus who develop NIP, management should include proactive monitoring and treatment of NIP with the goal of preserving mTOR inhibitor therapy.