The Drugs Don’t Work
 For those pop enthusiasts amongst you, “The drugs don’t work” – the year was 1997; The band: The Verve. For those more urologically minded, you will immediately be thinking of the recent publication in the Lancet reporting on the use of tamsulosin and nifedipine vs. placebo for the medical expulsive therapy (MET) of ureteric stones. Current national (BAUS) and international (EAU guidelines) recommend the use of MET, usually with an alpha blocker – and often tamsulosin, and it has certainly become common practice in most Emergency and urology departments certainly across the UK and likely worldwide.
For those pop enthusiasts amongst you, “The drugs don’t work” – the year was 1997; The band: The Verve. For those more urologically minded, you will immediately be thinking of the recent publication in the Lancet reporting on the use of tamsulosin and nifedipine vs. placebo for the medical expulsive therapy (MET) of ureteric stones. Current national (BAUS) and international (EAU guidelines) recommend the use of MET, usually with an alpha blocker – and often tamsulosin, and it has certainly become common practice in most Emergency and urology departments certainly across the UK and likely worldwide.
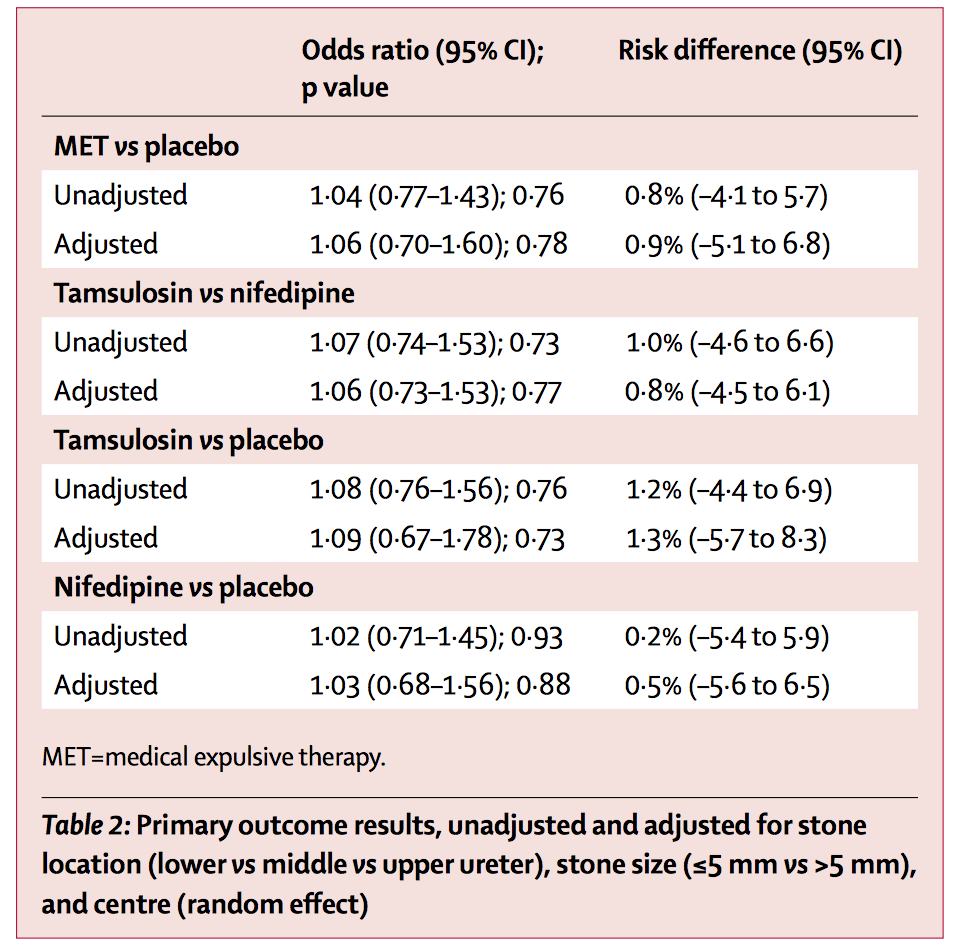
There have however always been doubts regarding the use of these with many small heterogenous studies with variable inclusion/exclusion criteria, various blinding protocols and suspicion of publication bias when only positive trials get published. Regardless, the clear outcome from the Hollingsworth meta-analysis was that both alpha blockers and calcium channel blockers are effective for helping stone passage and so they crept into routine clinical use. This trial should change all of that with headline results:
- No change in spontaneous stone passage at 4 weeks for either drug vs. placebo or compared against each other
- No difference when analysed by stone size or location
- No difference in analgesic use or time to stone passage
Aren’t those all the reasons we prescribe them? The first question of course is whether this trial is accurate. It certainly is a large trial with 1167 patients, randomised to the three double-blind arms in 24 centres in the UK. The trial (like many modern studies) is described as pragmatic. This has pros and cons. The advantages are that it replicates real life clinical practice allowing for variations in decision making (e.g. follow-up imaging in this paper) thus making it generally applicable. The downside of course is the lack of precision that this can introduce with stone passage possibly being only patient reported, or based on ultrasound, plain x-ray, IVU or CT. I guess we have to decide which type of trial we prefer, although it would be very difficult to mandate CT follow-up, with concerns about radiation safety, in this trial if that isn’t part of routine practice. Thus maybe this study is actually applicable to the vast amount of units around the world.
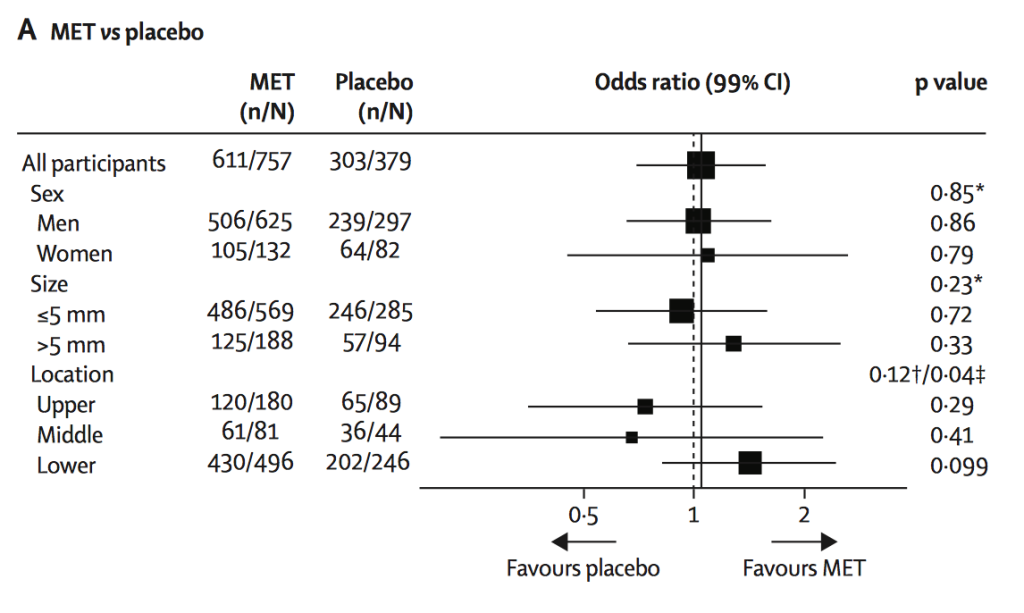
Secondly did it include the right type of patient? Well the current guidelines suggest using MET for any stone measuring up to 1cm in any part of the ureter … and that is what this trial did. And this is thus a strength given that it didn’t just focus on the distal ureter. Thus the trial population seems reasonable. One possibility is that if MET only works in the distal ureter (as almost all the studies only look at this), this could this explain the negative findings. Sub-group analysis of this based on location or stone size seems to suggest not unless it was underpowered to show a difference for this cohort of patients.
Whilst the odds ratios (see table) seem clear, the Forest plot shows the breakdown of subgroup by sex, stone size and location. Whilst not statistically significant, this does suggest a trend towards favouring MET for lower ureteric stones.
Thirdly, is it possible this trial is wrong … a type II error ? Well of course anything is possible, and the trial may be criticised for the follow-up mentioned above. However it does seem to provide easily the best evidence to date. Thus why has the use of MET been allowed to become routine practice based on a number of small trials all introducing inherent bias which is then amplified when a meta-analysis is performed. I guess it was the best evidence around at the time although it makes you wonder how many other interventions there are that we currently use that are based on smallish trials, and would they actually stand up to the rigour of a well conducted big multicentre trial?
My last question is will this change practice again? Well it should, but with no alternative (except time, fluids and NSAID’s) to offer patients with ureteric stones and given that alpha blockers are usually well-tolerated, I wonder whether people will continue to prescribe MET for the foreseeable future. But if we believe in evidence based medicine, and we do, then surely we should no longer prescribe MET for ureteric stones which after all is an off-licence indication.
Finally congratulations must go to the NIHR and the research team for answering a very important clinical question. Was the whole ‘MET’ story a placebo effect all along … or to quote another less well known song title from The Verve, was it “All in the mind”? The conclusion from this excellent study has to be yes.
Conflict of interest: Acted as PI for Guy’s and St. Thomas’ Hospital for this trial recruiting patients although have no part of study design, data analysis or publication.
Matthew Bultitude
Consultant Urologist, Guy’s and St. Thomas’ Hospital
Associate Editor, BJUI



Fantastic study. I am sure that there will be some criticisms but it seems very hard to get away from the high level of evidence and the convincing lack of benefit. Surely the end for MET?
Thanks Matt for a succinct and clear summary of a long-awaited paper.
You have to ask whether meta-analysed poor quality data or single high-quality RCT data are more important. In my opinion when it comes to MET the early data is weak enough that this robust study will change my practice.
There are many Endourologists using MET for other even less-well evidenced indications: fragments post SWL, post retrograde intra renal stone surgery etc.; will these practices now also reduce or stop? Do we need well-designed RCTs to answer these questions as well?! Perhaps we do, but in the meantime I’ll get back to re-writing our departmental guidelines.
COI- also PI for SUSPEND study, but no involvement in study design or write up
Love the music but not the drugs!
Very insightful comments by Matthew Bultitude. As practicing urologists that believe in evidence based medicine, the SUSPEND trial results should change our practice from today. MET doesn’t work…period!
… and if it does not work, then let’s disinvest and redirect NHS funds to alternative treatment options that work.
James N’Dow & Sam McClinton
Excellent discussion, Matt. It seems much easier to accept positive results than negative ones. Even with the subgroup analysis you quote suggesting a trend towards efficacy for distal ureteric stones, the power to detect a difference was very high, much higher than in any other study, and it was still negative.
As you mention, some urologists and emergency physicians will continue to prescribe alpha blockers in an effort to offer something other than analgesia and time. Perhaps we should go back to prescribing a ‘tincture of time’.
What do you think should be the next question we address for renal colic?
Conflict of interest: on the Steering Committee for the SUSPEND trial.
Superb summary of an outstanding paper – one of the best designed, executed and reported papers I have read in a long time. Whilst I look forward to discussing this paper for June’s #iurojc, the data convinces me to cease MET with immediate effect. EBM. Congratulations to the P.I.s and authors for an excellent piece of research.
COI: none.
Rustom Manecksha,
Endourologist, Dublin
Excellent commentary Matt, thank you. May I suggest ‘Time is a healer’ by Eva Cassidy.
Can anyone suggest an alternative placebo? It’s not easy getting these patients out of the Emergency Department.
Why don’t you take a rigid ureteroscope and stent down to the Emergency Department with you to show them!
Isn’t this a reiteration of already known facts ? (68% spontaneous expulsion for stones <5 mm as per EAU guidelines.)
In the present RCT, 75% of the stones were 5 mm and were comparable. The Forest plot shows a trend to support MET for calculus >5 mm and for lower ureteric stones, although such an assumption would be immature based on the provided data.
In addition to that the follow up criteria for the study is not based on repeat imaging, but based on symptomatic improvement and other factors.
We would like to have more discussions and enlightenment before making this the dogma
We should try to define the patients less likely to pass their stone spontaneously and treat them sooner.This study partly confirms some of that group.
Emergency ESWL within 48 hours of presentation for ureteric stones, particularly if larger and in the upper ureter, is a non-operative alternative for those with access to such a treatment. It is very acceptable to most patients and possibly more so than waving a ureteroscope and stent at them in the Emergency Department to convince them of a conservative approach.
However, recruiting patients to a trial of conservative management, lithotripsy or ureteroscopy is difficult for such different treatment options.
As a urologist with a 9mm upper ureteric stone who would want to try early emergency ESWL as a first option?
Continuing Matt Bultitude’s musical theme, the South African group Prime Circle produced an album entitled ‘Evidence’ in 2012 which included tracks entitled ‘Given The Truth’ & ‘Change’. With respect to medical expulsive therapy we have what is as close to the truth as we can hope for with the publication of this paper and it is now time to change. In the Hollingsworth meta-analysis cited in the blog, the largest study of the nine included in the analysis had 48 patients in each arm of the study. The Lancet paper discussed in this blog included 742 patients randomised with stones in the lower third of the ureter and 854 patients with stones of 5mm or less. Indeed, the total number of patients included in the Hollingsworth meta-analysis was 693, compared to 1167 patients in the present study. Prime Circle have two other tracks on the same album which seem appropriate to mention. One reminds me of the meta-analysis – ‘Written in Riddles’ whilst the other is simply entitled ‘Answers’. Sam McClinton and his team of investigators have provided answers to the question posed. It is right for us as clinicians to heed the lessons of this study and to change practice accordingly unless an equally well conducted and equally powered study should come along and suggest otherwise. Alpha-blockers and nifedipine don’t work in helping stones pass – move on.