We present an unusual case of unilateral simultaneous renal epithelioid angiomyolipoma (EAML) and renal clear cell carcinoma.
Authors: Qing-hua CAO 1, Fang LIU 2 , Ping XIAO 1, Xiao-ying TIAN 3 , Zhi LI 1* , Bin Li 1
1. Department of Pathology, The First Affiliated Hospital, Sun Yat-sen University
58, Zhongshan Road II, Guangzhou 510080, China
2. Cancer Center, Nan Fang Hospital of Southern Medical University
1838, Baiyun Av. North, Guangzhou 510515, China
3. Centre for Cancer and Inflammation Research, School of Chinese Medicine, Hong Kong Baptist University. 7, Baptist University Road, Kowloon Tong, Hong Kong, CHINA
Corresponding Author: Dr. Zhi LI, Associate professor. Department of Pathology, The First Affiliated Hospital, Sun Yat-sen University, 58, Zhongshan Road II, Guangzhou 510080, CHINA. E-mail: [email protected]
Abstract
A 52-year old male without sign of tuberous sclerosis had a complaint of 6-month history of pain in left renal area and had macroscopic hematuria twice within the preceding month. Computed tomography (CT) showed the presence of two masses in the upper and lower portions of the left kidney. Histological examination revealed the upper mass was composed of medium to large epithelioid cells with clear or eosinophilic cytoplasm and numerous giant multinucleated cells. Adult-appearing adipose tissue and coagulative necrosis could also be observed focally in the mass. Immunohistochemically, the tumour cells in the upper mass showed positive reactions to actin, HMB-45, Melan-A and CD68 but negative to pan-cytokeratin (CK), epithelial membrane antigen (EMA) and CD10. However, the lower mass was composed of diffusely monomorphic clear cells, strongly immunoreactive for pan-CK, vimentin, and CD10, without expression of HMB-45 and actin. To our knowledge, this is the first published report of coincidental malignant renal EAML and clear cell carcinoma in a kidney. Unlike conventional angiomyolipoma, adjuvant therapy after resection should be considered for renal EAML because of its malignant potential, more aggressive behaviour and poor prognosis.
Introduction
Conventional renal angiomyolipoma (AML) is a common tumour of the kidney, accounting for 2.0-6.4% of all renal tumours, and most of them are associated with a favourable prognosis [1-2]. Epithelioid angiomyolipoma (EAML) is a recently recognized rare variant of angiomyolipoma that is characterized by atypical histolo¬gical appearance with aggressive behaviour, mutations in the p53 gene and a high rate of distant metastases [3-4]. More than half of EAML in kidneys is associated with tuberous sclerosis (TS) [3, 5]. In rare cases, AML may occur together with renal cell neoplasm, and a retrospective study on 36 patients with concurrent AML and renal cell neoplasms has been described in the English scientific literature [6]. Herein, we report a case of coexistence of renal AML and renal clear-cell carcinoma occurring in a middle-aged man without TS. In contrast to previously-described unilateral simultaneous renal AML and renal clear-cell carcinoma, which exhibited histopathological features of conventional AML, our case presented polygonal epithelioid cells with nuclear atypia and prominent mitoses consistent with epithelioid angiomyolipoma. To our knowledge, this is the first report of concurrent epithelioid variant of AML and clear-cell carcinoma in the same kidney.
Case Presentation
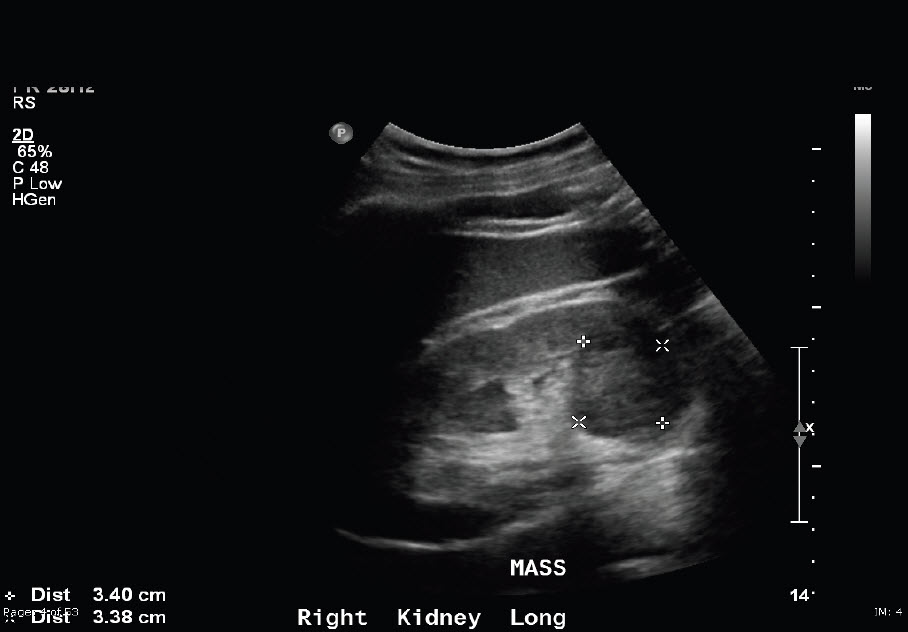
A 52-year-old male patient presented with a 6-month history of pain in left renal area and had macroscopic haematuria twice within the preceding month. On abdominal palpation, a non-tender left flank tumour was apparent. Ultrasonography and computerized tomography (CT) of the abdomen demonstrated two solid masses occupying two thirds of the upper and lower poles of the left kidney. The diameter of the longest part of the upper mass was approximately 14 cm, and the lower mass was approximately 4 cm in diameter (Figure 1).
Figure 1. Computerized tomography (CT) of the abdomen demonstrated two solid mass occupying the two thirds of upper middle portion of the left kidney (1) and lower pole of left kidney (2), respectively.
The left renal pelvis and calices were destroyed and adipose capsule surrounding renal was not able to be detected. The lesions radiographically were considered as suspicious for renal cell carcinoma. There was no clinical stigma of TS in either the patient or his family. The patient was admitted for excision of the mass and underwent an uneventful left radical nephrectomy.
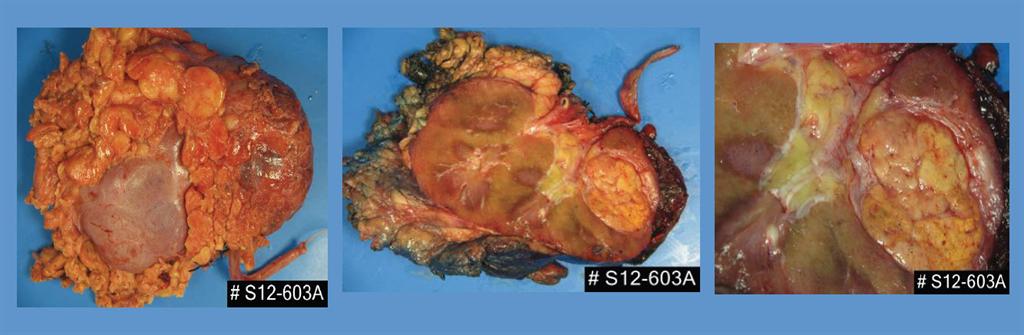
On macroscopical examination, the nephrectomy specimen was measured at 23×15×12cm. On the cut surface, normal structures of renal parenchyma were totally destroyed. Almost the whole left kidney was replaced by two solid masses. The upper mass was a 13.5×11.0×7.0cm brownish and soft tumour replacing two thirds of the upper middle portion of renal parenchyma. Haemorrhage and necrosis were observed in this mass. Adjacent to, but not continuous with, this larger upper mass was a 4.0×3.0×3.0 cm pale hard solid mass. The tumour was well circumscribed and located in the lower pole of the kidney. The mass was adherent to the subjacent renal capsule with gross involvement of the capsule (Figure. 2).
Figure 2. Gross findings for the two tumours in left kidney. The larger upper tumour had an expansive growth with marked necrosis (*). Cut surface was dark red in color (1).The lower tumour was well circumscribed and hard with pale colour. The tumour was adherent to the subjacent renal capsule with gross involvement of the capsule (2).

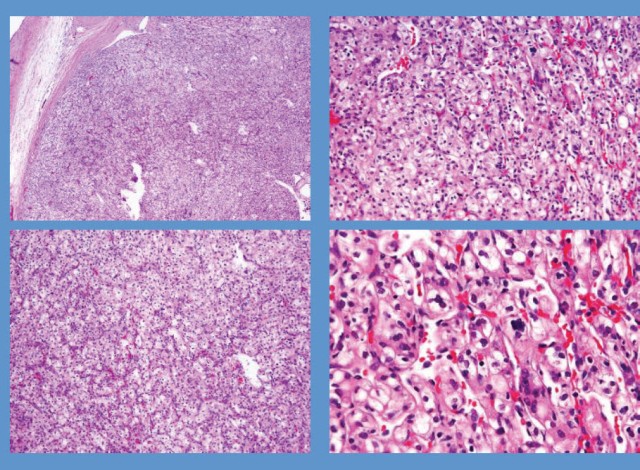
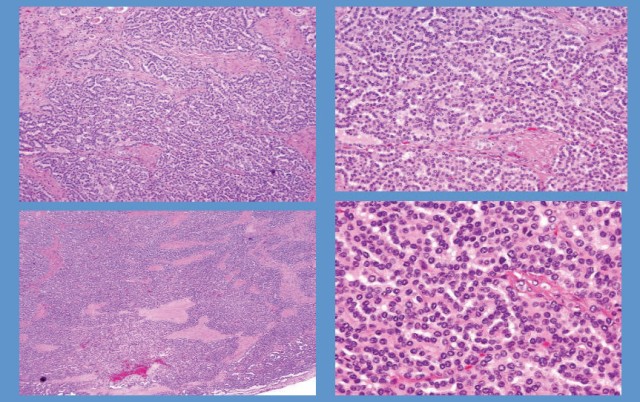
Microscopically, the upper tumour was composed of diffuse sheets of polygonal epithelioid cells, focally adult-appearing adipocytes and only scattered thick-walled blood vessels. The epithelioid cells had polymorphic and atypical nuclei with densely eosinophilic cytoplasm and prominent nucleoli similar to the ganglion cells. There were numerous multinucleated giant-cells. Mitotic figures were frequently observed (5/10 high-power fields) (Figure 3A). The spindle-shaped smooth muscle cells which frequently are present in conventional AML were not seen in this tumour. Haemorrhage and necrotic foci were also present. Renal capsular, vascular and lymphatic invasion were seen, but lymph nodes and the adrenal gland were negative for metastases (Figure 3B and 3C). The immunohistochemical staining showed that epithelioid cells were positive for HMB-45, Melan-A, actin and CD68; whereas they were negative for S-100, pan-cytokeratin, epithelial membrane antigen (EMA) and CD10 (Figure 3D). However, the histological appearance of the lower mass in the same kidney was completely different from the upper mass, in which alveolar architecture pattern with clear cells and delicate vascular network was noted. Coagulative necrosis and angioinvasion were not found in this lesion. The immunohistochemical staining showed that clear cells were strongly positive for vimentin, pan- cytokeratin and CD10, but negative for S-100, CD68, HMB-45, Melan-A, and actin (Figure 3E and 3F).
Figure 3. Photomicrographs of two tumours of left kidney. (A), the upper tumour showed pleomorphic and epithelioid tumour cells with large hyperchromatic nuclei and abundant eosinophilic cytoplasm (H&E, original magnification ×400). (B), in the upper tumour, the atypical epithelioid tumour cells invaded into the vascular lumen under the renal capsule (H&E, original magnification ×400). (C), in the upper tumour, abundant necrotic foci (*) could be observed (H&E, original magnification ×200). (D), in the upper tumour, the epithelioid tumour cells exhibited strong immunoreactivity with HMB-45 (immunohistochemical staining, original magnification ×400). (E), the lower tumour showed alveolar architecture pattern with clear cells and delicate vascular network (H&E, original magnification ×400). (F), in the lower tumour, the clear cells were diffusely positive for pan-CK (immunohistochemical staining, original magnification ×400).

Based on the above findings, the larger upper tumour of the left kidney was diagnosed as primary renal epithelioid angiomyolipoma, and the lower tumour was typical renal clear-cell carcinoma. The patient underwent left radical nephrectomy. The postoperative course was uneventful and no adjuvant therapy was given for the patient. The patient had been followed up for 1 year after surgery until he was lost to follow up. During the period of follow-up, no sign of recurrence and metastatic disease was found.
Discussion
Angiomyolipoma (AML) is the most common mesenchymal tumour of the kidney in adults, and now it is recognized as a member of the perivascular epithelioid clear cell (PEComa) family of tumours [7]. A common immunophenotype of these tumours is the consistent expression of melanoma-associated antigens, particularly HMB-45. AMLs are generally benign tumours because of their non-aggressive behaviour. Nodal involvements and renal vein extension in conventional AMLs are considered as multicentric occurrence but not metastasis [8-9]. Unlike conventional AMLs, renal epithelioid angiomyolipoma (EAML) is a rare, recently recognized variant of AML with more aggressive biological behaviour and marked cellular atypia. We reviewed the 22 cases of EAML reported in the past decade, and referred to the literature review from Sato et al [4]. It reveals the following facts: (a) More than one third of EAML patients have reported extension into the perirenal soft tissue and vena cava, and local recurrence and distant metastases to the lymph nodes, lungs and vertebrae. (b) The mean age of patients at diagnosis is 47 years (range 21 to 78 years), which is older than the patients with conventional AMLs (mean age 38 years). (c) EAMLs affect the genders in roughly equal numbers, whereas conventional AMLs predominantly occur in female subjects (4:1) in both sporadic and TS cases. (d) More than half of all patients diagnosed as EAMLs have an association with TS whose incidence is higher than that of conventional AMLs. Although 90% of renal AMLs are sporadic and non-associated with TS, approximately 50% of patients with TS can develop AML [10].TS-related AML is usually smaller, bilateral and multifocal according to Gomez criteria [11]. In our case, the patient had no obvious clinical manifestations of TS including seizures, mental retardation, or facial angiofibromas. The careful inspection of the nephrectomy specimen did not reveal additional foci of EAML. Therefore, we regarded the EAML in our case as sporadic. (e) Metastatic EAMLs usually have poor prognosis with more than 50% mortality, although a variety of chemotherapeutic agents have been applied to treat metastatic EAMLs because of their chemosensitivity [12]. In fact, long-term efficacy of chemotherapy for EAML remains to be determined. Histopathologically, it is difficult to predict the biological behaviour of EAML when there is absent evidence of distant metastasis. Some histological features such as necrosis, nuclear atypia, vascular invasion and mitotic activity have been suggested to predict the malignant behaviour of EAMLs [4, 13]. In our case, the tumour cells exhibited polymorphic and atypical nuclei with focal necrosis and vascular involvement. However, these histological features sometimes are inadequate to predict the prognosis of EAML patients because not all cases with cytologic atypia correlate with poor prognosis. Therefore, as a tumour with malignant potential and aggressive behaviour, we suggest that it is important to follow up those cases after surgery for recurrence or metastasis.
In rare cases, the simultaneous existence of AML and renal cell neoplasm can occur in the same kidney. Of a total reported 88 cases, clear cell renal-cell carcinoma was the most common histological subtype of renal cell neoplasm (62.5%, 55/88). Papillary renal-cell carcinoma was 4.5%(4/88), and urothelial carcinomas was 2.2%(2/88). The other rare histotypes of renal cell neoplasm included papillary adenoma (1), metanephric adenoma (2), chromophobe renal-cell carcinoma (4), and unclassified renal-cell carcinoma (11). Interestingly, oncocytoma was the second commonest histotype of renal cell neoplasm coexisting with AML (12.5%, 11/88). To our knowledge, so far, there are total 18 cases of concurrent AML and oncocytoma reported in the literature [14]. However, it is well known that oncocytomas account for only approximately 5% of surgically resected renal neoplasms in adults. The reason of why the relatively high incidence of oncocytoma in association with AML is still a matter of debate, and whether or not the coincidence of these two types of tumours are closely associated with TS remains to be determined. In the previous reports, we found that the histological types of all renal AMLs were conventional variants, and our presented case was the first report of coexistence of renal cell neoplasm with AML of epithelioid variant. However, it should be noted that most of previous cases were published before EAML was recognized, and some cases were later reclassified as an EAML rather than simultaneous existence of AML and clear-cell renal cell carcinoma [15]. Therefore, the true prevalence of coexistence of EAML with renal cell neoplasm including undetected cases may be higher than existing data.
Because of their epithelioid architectures histologically, some renal EAMLs may be misdiagnosed as renal cell carcinoma, renal sarcomas, or medullary carcinoma. EAML is distinguished from renal cell carcinoma due to lack of delicate vascular network and alveolar and tubular architectural patterns, all of which are characteristics of renal cell carcinoma. Immunohistochemically, renal cell carcinomas are positive for CK, EMA and CD10, which is not seen in the case of EAML. In contrast, the latter stains for HMB-45, smooth-muscle actin and sometimes CD68. Recently, CD117 was reported to be a useful marker for the diagnosis of angiomyolipoma as well. Renal sarcomas differ from EAML by their distinct morphoimmunophenotype. In the present case, the presence of foci of adult-appearing adipose tissue and scattered thick-walled blood vessels was helpful for getting the correct diagnosis. Medullary carcinomas are composed of cells arranged in solid nests or irregular tubules, but lack the large cells resembling ganglion cells and multinucleate giant cells. Moreover, positive immunostaining for HMB-45, actin, and CD68, and negative for epithelial markers were helpful for the EAML diagnosis.
Herein we reported an unusual case of unilateral coexistence of renal EAML with renal clear-cell carcinoma in same kidney. The EAML exhibited classical histological appearance and immunophenotype of angiomyolipoma with remarkable malignant features, including polymorphic and atypical nuclei, focal necrosis and vascular involvement, which was not frequently described in the previous similar reports. EAML is a rare entity with aggres¬sive behaviour and poor prognosis. Despite the lack of distant metastasis in our case during the period of follow-up, we postulate its malignant potential and suggest a long period of follow-up is necessary. Adjuvant therapy after resection should be considered for renal EAML. That will help establish a standard for treatment and follow-up of EAML.
Statements: no conflict of interest or financial support declared.
Reference
1. Lopez-Beltran A, Scarpelli M, Montironi R et al. 2004 WHO classification of the renal tumors of the adults. Eur Urol, (2006) 49:798-805.
2. Martigoni G, Amin MB: Angiomyolipoma, in Eble JN, Sauter G, Epstein JI, Sesterhenn IA (Ed) World Health Organization classification of tumors. Pathology and genetics of tumors of the urinary system and male genital organs. IARC Press, Lyon(2004): 63-67.
3. Amin MB: Epithelioid angiomyolipoma, in: Eble I, Sauter JN, Epstein G, Sesterhenn JI, Health IA (Ed) World Health Organization classification of tumors. Pathology and genetics of tumors of the urinary system and male genital organs. IARC Press, Lyon (2004): 68-69.
4. Sato K, Ueda Y, Tachibana H, Miyazawa K et al. Malignant epithelioid angiomyolipoma of the kidney in a patient with tuberous sclerosis: An autopsy case report with p53 gene mutation analysis. Pathol Res Pract, (2008)204: 771-777.
5. Bjornsson J, Short MP, Kwiattkowski DJ et al. Tuberous sclerosis associated renal cell car¬cinoma, clinical, pathological and genetic features. Am J Surg Pathol, (1996)149: 1201-1208.
6. Jimenez RE, Eble JN, Reuter VE et al. Concurrent angiomyolipoma and renal cell neoplasia: a study of 36 cases. Mod Pathol, (2001)14:157-163.
7. Hornick JL, Fletcher CD PEComa: What do we know so far? Histopathology, (2006) 48:75-82.
8. Bloom DA, Scardino PT, Ehrlich RM The significance of lymph nodal involvement in renal angiomyolipoma. J Urol, (1982) 128: 1292-1295.
9. Wilson SS, Clark PE, Stein JP Angiomyolipoma with vena caval extension. Urology, (2002) 60: 695-696.
10. Bissler JJ, Kingswood JC Renal angiomyolipomata. Kidney Int, (2004)66:924-934.
11. Gomez MR. Phenotypes of the tuberous sclerosis complex with a revision of diagnostic criteria. Ann N Y Acad Sci, (1991) 615:1-7.
12. Park HK, Zhang S, Wong MK et al. Clinical presentation of epithelioid angiomyolipoma. Int J Urol, (2007)14:21-25.
13. Kawaguchi K, Oda Y, Nakanishi K et al. Malignant transformation of renal angiomyolipoma. A case report. Am J Surg Pathol, (2002)26:523-529.
14. Bahrami A, Schwartz MR, Ayala AG et al. Concurrent angiomyolipoma and two oncocytomas in the same kidney. Ann Diag Pathol, (2007) 11:132-136.
15. Pea M, Bonetti F, Martignoni G et al. Apparent renal cell carcinomas in tuberous sclerosis are heterogeneous: the identification of malignant epithelioid angiomyolipoma. Am J Surg Pathol, (1998) 22:180-187.
16. Makhlouf HR, Remotti HE, Ishak KG et al. Expression of KIT (CD117) in angiomyolipoma. Am J Surg Pathol, (2002) 26: 493-497.
17. Camporo P, Vasiliu V, Molinie V et al . Renal translocation carcinomas. Clinicopathologic, immunohistochemical, and a gene expression profiling analysis of 31 cases with a review of literature. Am J Surg Pathol, (2008)35:656-670.
Date added to bjui.org: 28/07/2011
DOI: 10.1002/BJUIw-2011-038-web