Article of the Week: Association between T2DM, curative treatment and survival in localized PCa
Every Week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
Finally, the third post under the Article of the Week heading on the homepage will consist of additional material or media. This week we feature a video discussing the paper.
If you only have time to read one article this week, it should be this one.
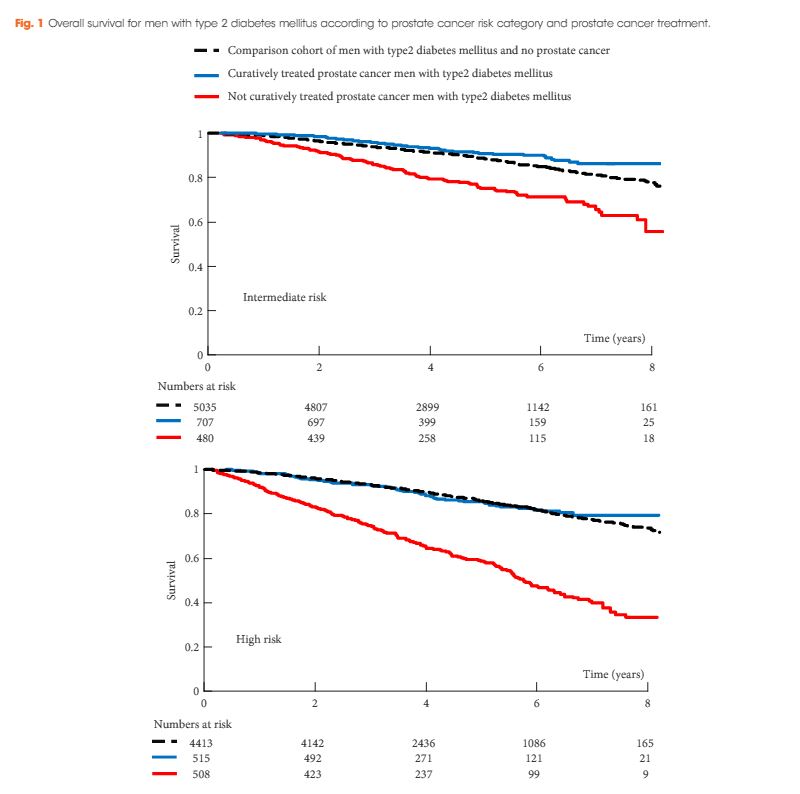
Association between type 2 diabetes, curative treatment and survival in men with intermediate- and high-risk localized prostate cancer
Abstract
Objective
To investigate whether curative prostate cancer (PCa) treatment was received less often by men with both PCa and Type 2 diabetes mellitus (T2DM) as little is known about the influence of T2DM diagnosis on the receipt of such treatment in men with localized PCa.
Subjects and Methods
The Prostate Cancer database Sweden (PCBaSe) was used to obtain data on men with T2DM and PCa (n = 2210) for comparison with data on men with PCa only (n = 23 071). All men had intermediate- (T1–2, Gleason score 7 and/or prostate-specific antigen [PSA] 10–20 ng/mL) or high-risk (T3 and/or Gleason score 8–10 and/or PSA 20–50 ng/mL) localized PCa diagnosed between 1 January 2006 and 31 December 2014. Multivariate logistic regression was used to calculate the odds ratios (ORs) for receipt of curative treatment in men with and without T2DM. Overall survival, for up to 8 years of follow-up, was calculated both for men with T2DM only and for men with T2DM and PCa.
Results
Men with T2DM were less likely to receive curative treatment for PCa than men without T2DM (OR 0.78, 95% confidence interval 0.69–0.87). The 8-year overall survival rates were 79% and 33% for men with T2DM and high-risk PCa who did and did not receive curative treatment, respectively.
Conclusions
Men with T2DM were less likely to receive curative treatment for localized intermediate- and high-risk PCa. Men with T2DM and high-risk PCa who received curative treatment had substantially higher survival times than those who did not. Some of the survival differences represent a selection bias, whereby the healthiest patients received curative treatment. Clinicians should interpret this data carefully and ensure that individual patients with T2DM and PCa are not under- nor overtreated.