Posts
Video: Step-By-Step: Extended PLND – Creating the Spaces
Sequencing robot-assisted extended pelvic lymph node dissection prior to radical prostatectomy: a step-by-step guide to exposure and efficiency
OBJECTIVE
To describe a novel, step-by-step approach to robot-assisted extended pelvic lymph node dissection (ePLND) at the time of robot-assisted radical prostatectomy (RARP) for intermediate–high risk prostate cancer.
PATIENTS AND METHODS
The sequence of ePLND is at the beginning of the operation to take advantage of greater visibility of the deeper hypogastric planes. The urachus is left intact for an exposure/retraction point. The anatomy is described in terms of lymph nodes (LNs) that are easily retrieved vs those that require additional manipulation of the anatomy, and a determined surgeon. A representative cohort of 167 RARPs was queried for representative metrics that distinguish the ePLND: 146 primary cases and 21 with neoadjuvant systemic therapy.
RESULTS
The median (interquartile range, IQR) LN yield was 22 (16–28) for primary surgeries and 21 (16–23) for neoadjuvant cases. The percentage of cases with positive LNs (pN1) was 16.4% for primary and 29% for neoadjuvant. The hypogastric LNs were involved in 75% of pN1 primary cases and uniquely positive in 33%. Each side of ePLND took the attending surgeon a median (IQR) of 16 (13–20) min and trainees 25 (24–38) min.
CONCLUSIONS
Robot-assisted ePLND before RARP provides an anatomical approach to surgical extirpation mimicking the open approach. We think this sequence offers efficiency and efficacy advantages in high-risk and select intermediate-risk patients with prostate cancer undergoing RARP.
Article of the Week: Predicting pathological outcomes in patients undergoing RARP for high-risk prostate cancer
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
Finally, the third post under the Article of the Week heading on the homepage will consist of additional material or media. This week we feature a video from Dr. Firas Abdollah, discussing his paper.
If you only have time to read one article this week, it should be this one.
Predicting Pathologic Outcomes in Patients Undergoing Robot-Assisted Radical Prostatectomy for High Risk Prostate Cancer: A Preoperative Nomogram
OBJECTIVE
To identify which high-risk patients with prostate cancer may harbour favourable pathological outcomes at radical prostatectomy (RP).
PATIENTS AND METHODS
We evaluated 810 patients with high-risk prostate cancer, defined as having one or more of the following: PSA level of >20 ng/mL, Gleason score ≥8, clinical stage ≥T2c. Patients underwent robot-assisted RP (RARP) with pelvic lymph node dissection, between 2003 and 2012, in one centre. Only 1.6% (13/810) of patients received any adjuvant treatment. Favourable pathological outcome was defined as specimen-confined disease (SCD; pT2–T3a, node negative, and negative surgical margins) at RARP-specimen. Logistic regression models were used to test the relationship among all available predicators and harbouring SCD. A logistic regression coefficient-based nomogram was constructed and internally validated using 200 bootstrap resamples. Kaplan–Meier method estimated biochemical recurrence (BCR)-free and cancer-specific mortality (CSM)-free survival rates, after stratification according to pathological disease status.
RESULTS
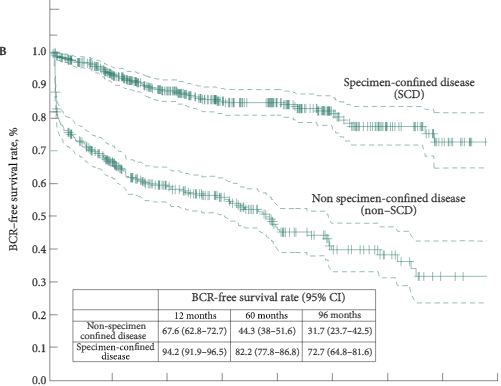
Overall, 55.2% patients harboured SCD at RARP. At multivariable analysis, PSA level, clinical stage, primary/secondary Gleason scores, and maximum percentage tumour quartiles were all independent predictors of SCD (all P < 0.04). A nomogram based on these variables showed 76% discrimination accuracy in predicting SCD, and very favourable calibration characteristics. Patients with SCD had significantly higher 8-year BCR- (72.7% vs 31.7%, P < 0.001) and CSM-free survival rates (100% vs 86.9%, P < 0.001) than patients with non-SCD.
CONCLUSIONS
We developed a novel nomogram predicting SCD at RARP. Patients with SCD achieved favourable long-term BCR- and CSM-free survival rates after RARP. The nomogram may be used to support clinical decision-making, and aid in selection of patients with high-risk prostate cancer most likely to benefit from RARP.
Editorial: More Nomograms or Better Lymph node dissection – What do we need in Prostate Cancer?
The publication of nomograms to predict radical prostatectomy (RP) outcome using preoperative parameters were important steps in urological oncology. Abdollah et al. [1], in this issue of BJU International, present a new nomogram to predict specimen-confined disease (SCD; pT2–3a, pN0 R0) in men with high-risk prostate cancer undergoing pelvic lymph node dissection (PLND) and robot-assisted RP (RARP). They used statistical logistic regression to measure the impact of various preoperatively available clinicopathological parameters on the likelihood of pathological outcome and tumour recurrence. The final nomogram accurately identified SCD (pT2–3a, pN0 R0) in 76% of the patients. It is intuitive that these patients have good long-term oncological outcomes after surgery. Consequently, Abdollah et al. found excellent 8-year cancer-specific survival rates in these patients. Because nomograms provide individualised risk prediction for patients in an easily applicable manner, they have become very popular among clinicians. Nomograms are now being applied for almost every aspect of prostate cancer. These are freely available and both patients and physicians are encouraged to use them.
Although nomograms undoubtedly have improved our perspective of disease behaviour and individual patient prediction, several key questions remain. First, how good are the input data to a nomogram? Abdollah et al. [1] evaluated 810 patients with high-risk prostate cancer treated in a single large centre between 2003 and 2012. Impressively, more than half of the patients (55%) harboured SCD at RARP. Such a high chance of having SCD will probably encourage many physicians and patients to choose surgery, even without using a nomogram, because this approach may avoid the need for hormonal treatment, which is obligatory for radiation therapy in high-risk prostate cancer. Second, is the predictive accuracy safe within clinical practice? Most nomograms using clinicopathological data generate predictive accuracies within the range of 75–90% (including the nomogram presented by Abdollah et al. [1]). It is of special importance to consider that 64/447 (14%) of the patients with SCD in the series reported by Abdollah et al. [1] received salvage treatment, which was initiated after a median (interquartile range, IQR) of 4.8 (1.4–9.3) months, and the indication to initiate this salvage therapy was PSA recurrence. Obviously, these patients did not have specimen confined disease and were misclassified. In this case, one might postulate a persistence of nodal disease, given an inadequate extent of PLND. Abdollah et al. [1] reported on a median (IQR) of 5 (3.0–11.0) lymph nodes removed.
In their landmark paper on extended PLND (ePLND) in cadavers, Weingartner et al. [2] demonstrated that a mean lymph node yield of 20 serves as a guideline for sufficient ePLND. More than 10 years ago, Heidenreich et al. [3] reported on a 15% higher rate of lymph node metastasis detection when comparing ePLND with the standard LND (obturator). Bader et al. [4] provided further evidence that an ePLND is needed to provide adequate clinical staging and potential therapeutic benefit. Of 365 patients with clinically localised prostate cancer, 88 (24%) had positive lymph nodes. In this series, a pelvic LND that spared the internal iliac bed would have left 58% of patients with positive nodes with residual disease and 19% would have been incorrectly staged as lymph node-negative for cancer. These data were recently confirmed by several authors when analysing retrospective series. Furthermore, Seiler et al. [5] updated their series of 88 patients and recently reported on the long-term outcome after a median follow-up of 15.6 years. They showed that 18% of those patients with one positive node remained biochemical recurrence free, 28% showed biochemical recurrence only, and 54% had clinical progression. Of these 39 patients, 57% never required deferred androgen-deprivation therapy. In contrast, patients with multiple positive nodes are likely to experience rapid progression and, thus, may benefit from early adjuvant therapies. International clinical practice guidelines recommend the performance of an anatomically ePLND at RP in men with high-risk prostate cancer, for both staging and therapeutic purposes.
Nowadays, most urologists claim to perform an ePLND. However, a recent analysis among 50 671 men who were surgically treated with RP from 2010 to 2011 in the USA showed that, overall, only 69.3% of the high-risk patients underwent concomitant PLND [6]. Surgical approach and hospital characteristics were associated with treatment with PLND and detection of lymph node metastasis. More specifically, patients with prostate cancer undergoing open RP or surgically treated at high-volume centres were more likely to undergo PLND than those undergoing RARP or surgically treated at low-volume centres.
Despite the strong evidence that ePLND positively affects survival in men with limited lymph node involvement, this procedure is not commonly performed. The reasons for this are multiple and include expertise, stage migration and functional and oncological outcomes, as well as economics and the introduction of laparoscopic and laparoscopic RARP. However, this is no reason not to offer the patient, if possible, an operation which has the highest chance of cure
Video: Predicting pathological outcomes in patients undergoing RARP for high-risk prostate cancer: A Preoperative Nomogram
Predicting Pathologic Outcomes in Patients Undergoing Robot-Assisted Radical Prostatectomy for High Risk Prostate Cancer: A Preoperative Nomogram
OBJECTIVE
To identify which high-risk patients with prostate cancer may harbour favourable pathological outcomes at radical prostatectomy (RP).
PATIENTS AND METHODS
We evaluated 810 patients with high-risk prostate cancer, defined as having one or more of the following: PSA level of >20 ng/mL, Gleason score ≥8, clinical stage ≥T2c. Patients underwent robot-assisted RP (RARP) with pelvic lymph node dissection, between 2003 and 2012, in one centre. Only 1.6% (13/810) of patients received any adjuvant treatment. Favourable pathological outcome was defined as specimen-confined disease (SCD; pT2–T3a, node negative, and negative surgical margins) at RARP-specimen. Logistic regression models were used to test the relationship among all available predicators and harbouring SCD. A logistic regression coefficient-based nomogram was constructed and internally validated using 200 bootstrap resamples. Kaplan–Meier method estimated biochemical recurrence (BCR)-free and cancer-specific mortality (CSM)-free survival rates, after stratification according to pathological disease status.
RESULTS
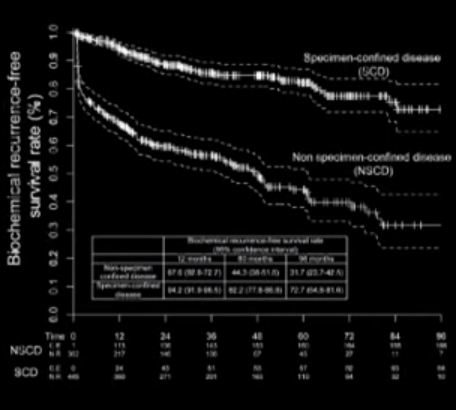
Overall, 55.2% patients harboured SCD at RARP. At multivariable analysis, PSA level, clinical stage, primary/secondary Gleason scores, and maximum percentage tumour quartiles were all independent predictors of SCD (all P < 0.04). A nomogram based on these variables showed 76% discrimination accuracy in predicting SCD, and very favourable calibration characteristics. Patients with SCD had significantly higher 8-year BCR- (72.7% vs 31.7%, P < 0.001) and CSM-free survival rates (100% vs 86.9%, P < 0.001) than patients with non-SCD.
CONCLUSIONS
We developed a novel nomogram predicting SCD at RARP. Patients with SCD achieved favourable long-term BCR- and CSM-free survival rates after RARP. The nomogram may be used to support clinical decision-making, and aid in selection of patients with high-risk prostate cancer most likely to benefit from RARP.