Article of the Week: Prostatic urethral lift vs transurethral resection of the prostate
Every week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
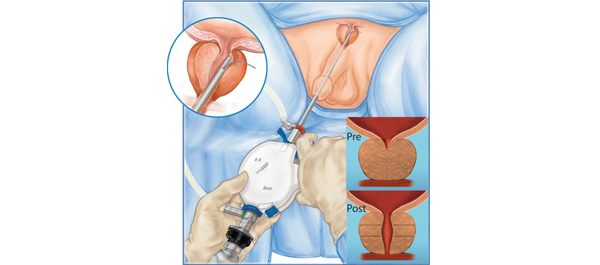
Prostatic urethral lift vs transurethral resection of the prostate: 2-year results of the BPH6 prospective, multicentre, randomized study
Abstract
Objectives
To compare prostatic urethral lift (PUL) with transurethral resection of the prostate (TURP) with regard to symptoms, recovery experience, sexual function, continence, safety, quality of life, sleep and overall patient perception.
Patients and Methods
A total of 80 patients with lower urinary tract symptoms attributable to benign prostatic hyperplasia (BPH) were enrolled in a prospective, randomized, controlled, non-blinded study conducted at 10 European centres. The BPH6 responder endpoint assessed symptom relief, quality of recovery, erectile function preservation, ejaculatory function preservation, continence preservation and safety. Additional evaluations of patient perspective, quality of life and sleep were prospectively collected, analysed and presented for the first time.
Results
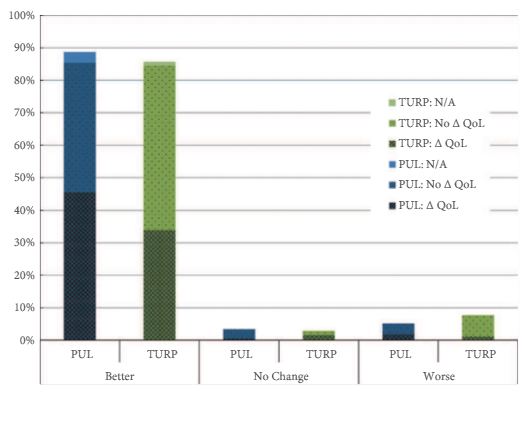
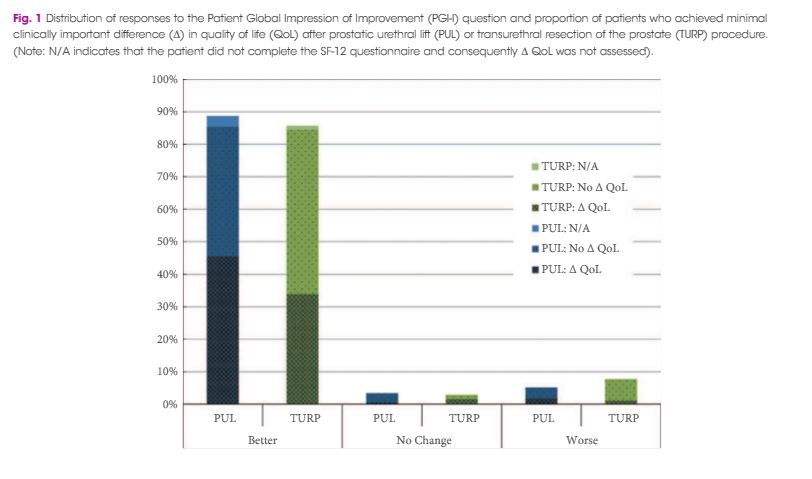
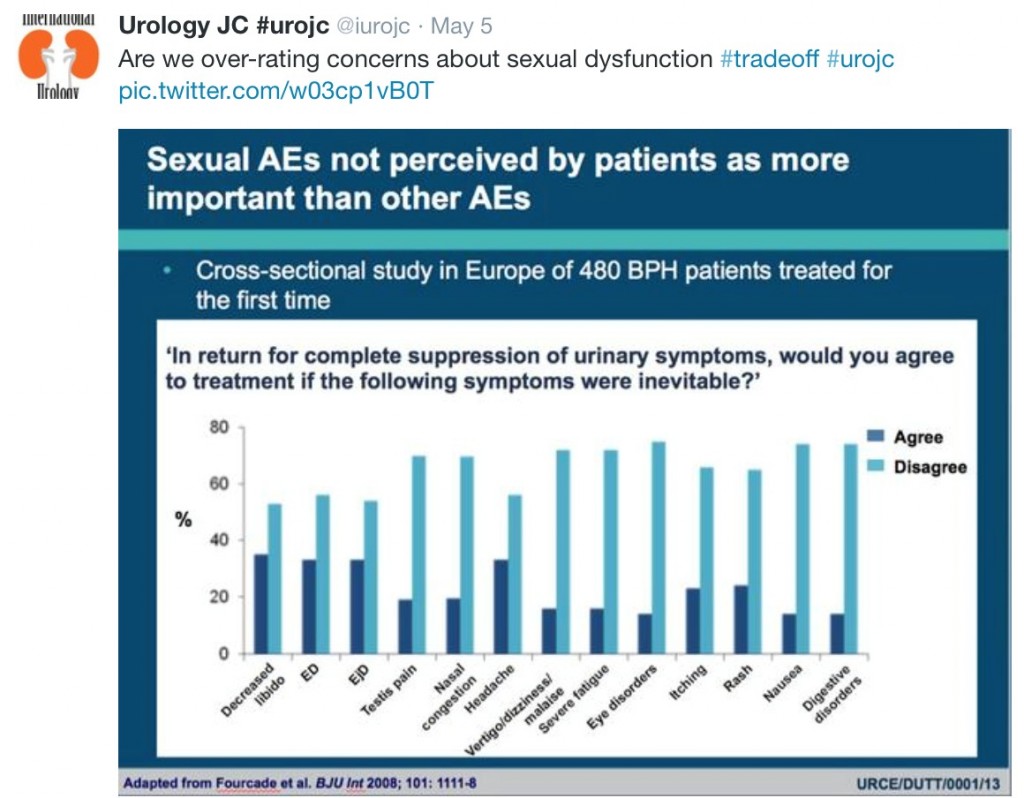
Significant improvements in International Prostate Symptom Score (IPSS), IPSS quality of life (QoL), BPH Impact Index (BPHII), and maximum urinary flow rate (Qmax) were observed in both arms throughout the 2-year follow up. Change in IPSS and Qmax in the TURP arm were superior to the PUL arm. Improvements in IPSS QoL and BPHII score were not statistically different between the study arms. PUL resulted in superior quality of recovery, ejaculatory function preservation and performance on the composite BPH6 index. Ejaculatory function bother scores did not change significantly in either treatment arm. TURP significantly compromised continence function at 2 weeks and 3 months. Only PUL resulted in statistically significant improvement in sleep.
Conclusion
PUL was compared to TURP in a randomised, controlled study which further characterized both modalities so that care providers and patients can better understand the net benefit when selecting a treatment option.