2 Department of Radiology, Royal Melbourne Hospital, Melbourne, Victoria, Australia
It is rare for embryologic abnormalities of the urinary tract to present in adulthood, especially as an incidental finding post radical perineal prostatectomy (RPP). We present the incidental diagnosis and subsequent management of a 68 year old man with clinically localised prostate cancer and a complete duplication of the right collecting system associated with an asymptomatic upper pole ectopic ureter, that was inadvertently injured during open RPP.
With the earlier detection of clinically localised prostate cancer using prostate specific antigen (PSA) testing, it has become routine not to perform extensive staging investigations to avoid unnecessary tests. This approach may lead to rare unforeseen post-operative complications in patients with asymptomatic congenital anomalies of the urinary tract. We report the case of a 68 year old man who presented with persistent urinary extravasation from his surgical wound following RPP. Subsequent CT intravenous urogram demonstrated an incidental complete duplication of the right collecting system and an injured right upper pole ectopic ureter. To our knowledge, there are only four cases reported in the literature of ectopic ureters found incidentally in men undergoing radical prostatectomy for prostate cancer (1-4), and we describe the first case reported during RPP.
A 68 year old asymptomatic man initially presented with an incidental elevated PSA of 4.8 ng/ml (12% free) detected on routine testing. The patient otherwise had no significant past medical history. On digital rectal examination, the prostate was mildly enlarged and firm but with no palpable nodules bilaterally. Trans-rectal ultrasound (TRUS) of the prostate with concurrent 12 core guided biopsies, revealed no suspicious areas and an estimated prostate volume of 60 cm3. Histopathology showed a 2mm focus of Gleason 6 (3+3) prostate adenocarcinoma, which was only detected following further immunohistochemical staining. An active surveillance approach with six monthly PSA tests was initially adopted given the very low volume of malignancy. Over the next 18 months, the PSA gradually rose to 8.2 ng/ml whereby a repeat TRUS guided biopsy revealed prostate adenocarcinoma with Gleason 6 (3+3) in 33% of 1 core obtained from the right lateral prostate (clinical stage T1c).
Following patient counselling, a radical nerve-sparing perineal prostatectomy was performed using a previously described technique (5). A routine pre-operative rigid cystoscopy revealed a normal urethra and bladder with two appropriately located ureteric orifices. Surgery went as planned and a 16Fr two-way silicone indwelling catheter was inserted into the bladder intra-operatively. The urethral anastomosis was subsequently checked to ensure a watertight seal and a tube drain was also inserted through a separate stab incision into the perineum. Pelvic lymphadenectomy was not performed due to the patient’s low risk pathology. The final pathology demonstrated a Gleason 7 (3+4) adenocarcinoma, localised predominantly to the right lower zone involving approximately 15% of total prostatic volume. Lymphovascular invasion was absent and the surgical margins were clear.
Early post-operative recovery was uneventful and the drain tube was removed on post-operative day (POD) 2. A routine trial of void 2 weeks post surgery resulted in a persistent urine leak from the apex of his perineal wound, which was exacerbated by urination. Of note, the patient remained pain free and afebrile throughout the post-operative period. This was initially suggestive of an anastomotic leak and therefore the indwelling urinary catheter was re-inserted. However, a cystogram did not show any extravasation of contrast (Figure 1) and the leak persisted through the apex of the wound, so the re-introduced urinary catheter was removed.
Figure 1. Post-operative retrograde cystogram showing no anastomotic leakage
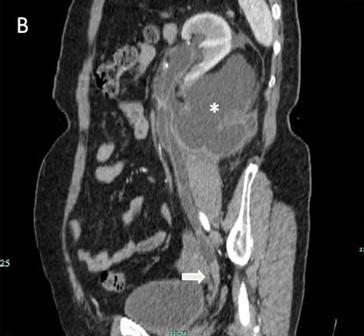
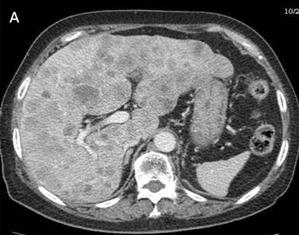
Subsequent computed tomography (CT) intravenous urogram demonstrated a complete duplication of the right collecting system with an ectopic right upper pole ureter (Figure 2a and 2b), the distal end of which could not be properly visualised.
Figure 2a. Coronal view of the CT intravenous urogram showing the right lower pole ureter (LPU) and the right upper pole ectopic ureter (UPEU).
Figure 2b. 3D reconstruction of the coronal CT-intravenous urogram showing the orthotopic insertion of the right lower pole ureter (LPU) into the bladder (solid arrow), the tortuous right upper pole ectopic ureter (UPEU, dotted arrow). Note an indwelling urinary catheter is in situ.
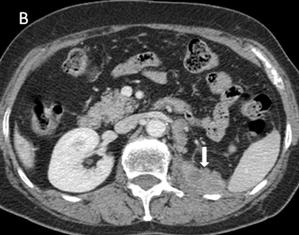
There was also a fistulous tract originating from the ectopic ureter, draining into the perineum (Figure 3). The patient subsequently developed right-sided epididymo-orchitis, which was successfully treated with intravenous gentamicin and amoxicillin.
Figure 3. Coronal view of the CT intravenous urogram demonstrating the right upper and lower pole renal moieties, as well as a fistulous tract (solid arrow) originating from the ectopic ureter into the perineum on the right side (dotted arrows).

Management
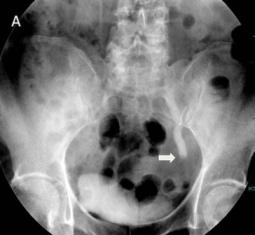
The patient subsequently underwent a rigid cystoscopy and a right retrograde pyelogram, which demonstrated a non-dilated right ureter draining into a lower renal moiety, producing a ‘drooping lily’ type image of the right kidney (Figure 4).
Figure 4. Right retrograde pyelogram demonstrating the characteristic ‘drooping lily’ image of the lower renal moiety.

After patient counselling, the decision was then made to perform an open right ureteric reimplantation. Intra-operatively, two ureters were identified in the retroperitoneum adjacent to the bifurcation of the right common iliac vessel. The lateral ureter was entered via a small stab incision and dye was injected. This dye was noted to be draining out of the indwelling catheter and therefore this was thought to be the normal ureter that was draining into the bladder. The medial ureter was also pierced and dye was injected, but the dye was not detected in the bladder. Thus, this was thought to be the ectopic ureter, most likely to have been injured during the RPP. Given the close proximity of the bladder to the ectopic ureter, a uretero-vesical anastomosis was performed. The distal ectopic ureter was spatulated and a cystotomy was made at a corresponding site. A 6Fr 26 cm ureteric stent was passed through the ectopic ureter over a guide wire up to the right kidney, and the distal end was placed through the cystotomy. Using four, 4-zero vicryl sutures in an interrupted fashion, the ureter was securely implanted onto the bladder with good mucosal apposition. A pelvic drain tube was placed adjacent to the newly formed uretero-vesical anastomosis. There were no peri-operative complications, the drain was removed on POD 4 and the patient was discharged home on POD 6. A flexible cystoscopy was performed approximately 4 weeks after to remove the ureteric stent. Repeat CT intravenous urogram performed 2 months post re-implantation demonstrated complete duplication of the right collecting system with double ureters in which contrast drained normally into the bladder. Importantly, it did not show any hydronephrosis bilaterally, no perineal collection nor any leak from the right renal tract (Figure 5).
Figure 5. Coronal view of the CT intravenous urogram post uretero-vesical anastomosis of the right upper pole ectopic ureter, showing resolution of the fistulous tract to the perineum.
Discussion
We report the case of a patient who was incidentally found to have a complete duplication of the right collecting system with an asymptomatic ectopic upper pole ureter that was inadvertently injured during a RPP. To our knowledge, this is the first case reported in the literature associated with such surgery.
A duplex system refers to a kidney with two pelvicalyceal systems within a single renal parenchyma, which may have either a single or bifid ureter (partial duplication), or two discrete ureters (complete duplication) (6). Renal duplication is a relatively common congenital anomaly, with a reported prevalence of 0.3-6% (7). The majority of cases are detected during childhood, but up to 20% of patients remain asymptomatic into adulthood (8). In contrast, ectopic ureters are rare, with a reported incidence of 1 in 1900 (9). Ectopic ureters are more common in females and are usually associated with a duplicated collecting system, whereas they are typically associated with a single collecting system in men (10).The anatomic site of insertion of an ectopic ureter in a duplicated collecting system follows the Weigert-Meyer rule, whereby the upper pole ureter is more commonly ectopic and the lower pole ureter typically inserts into the trigone, or laterally and cranially to this structure. In 50-60% of patients, renal duplication is associated with vesicoureteric reflux, which may affect one or both ureters. Reflux is more common in the lower pole kidney (97%), while ureteroceles and ectopic ureters are commoner in the upper pole kidney (11).
The presentation of ectopic ureters depends on their site of insertion. Ectopic ureters inserting into the prostatic urethra typically present with urinary tract infections or lower urinary tract symptoms of urgency and frequency. Ectopic ureters inserting into a seminal vesicle, vas deferens or epididymis may present with epididymitis, chronic prostatitis, abdominal or pelvic pain, discomfort during ejaculation, constipation, or a large abdominal mass secondary to obstruction and hydronephrosis. Males with an ectopic ureter may also present with infertility (3).
In males, the majority of ectopic ureters (50%) insert into the prostatic urethra, and 33% into a seminal vesicle. The least common sites include the prostatic utricle and the vas deferens (12). Ectopic ureters in males almost never insert distal to the external sphincter, where it would present as urinary incontinence. One such case has been reported in the literature (13). Of note, this patient had no lower urinary tract symptoms prior to the RPP. It is likely that in this case, the upper pole ectopic ureter was transected during the bladder neck dissection at the level of the prostatic urethra.
There are multiple treatment options for the pathological ectopic ureter associated with a duplicated system, including ureteropyelostomy, heminphrectomy, ipsilateral ureteroureterostomy and common sheath reimplantation (14). An ureterovesical anastomosis was performed in this case due to the upper pole ectopic ureter being a distinct entity from the lower pole ureter, and because of its close proximity to the bladder.
Current guidelines do not recommend imaging of the prostate with CT or MRI for low risk disease prior to radical prostatectomy. Consequently, any asymptomatic congenital anomalies of the urinary tract will not be detected preoperatively. However, due to the rarity of such anomalies, as well as the expense and potential risk to patients, routine preoperative imaging to detect congenital abnormalities is not justifiable in this setting. In the retrospective study conducted by Costa et al, only 3 out of 254 (1.1%) surgical descriptions of nephroureterectomy samples showed anatomical variations of the collecting system, with all cases demonstrating ureteric duplications (15).
Of interest, given the established genetic association of duplex systems, the patient’s twin brother subsequently underwent a CT intravenous urogram, which did not demonstrate any evidence of a duplicated system or ectopic ureter.
Conclusion
Preoperative imaging may help detect asymptomatic congenital abnormalities of the urinary tract, enabling appropriate intraoperative management. However, given the rarity of such anomalies and the expense of imaging, extensive investigations prior to radical prostatectomy for the sole purpose of screening for congenital abnormalities is not justifiable. However, this case highlights the need to consider congenital anomalies of the urinary tract as a possible differential in peri-operative complications post radical prostatectomy.
References
1. Funahashi Y, Kamihira O, Kasugai S, Kimura K, Fukatsu A, Matsuura O. [Radical prostatectomy for prostate carcinoma with ectopic ureter ; a case report]. Nihon Hinyokika Gakkai zasshi The japanese journal of urology. 2007 2007;98(3):580-2.
2. Ghazi A, Zimmermann R, Janetschek G. Delayed detection of injury to an ectopic ureter of a duplicated collecting system following laparoscopic radical prostatectomy for early organ-confined prostate cancer. Urologia internationalis. 2011 Feb (Epub 2010 Nov;86(1):121-4.
3. Marien TP, Shapiro E, Melamed J, Taouli B, Stifelman MD, Lepor H. Management of localized prostate cancer and an incidental ureteral duplication with upper pole ectopic ureter inserting into the prostatic urethra. Reviews in urology. 2008 2008;10(4):297-303.
4. Nakai Y, Tanaka M, Yoshikawa M, Tanaka N, Hirayama A, Fujimoto K, et al. [Prostate cancer and left ectopic ureter opening to seminal vesicle with left renal agenesis: a case report]. Hinyokika kiyo Acta urologica Japonica. 2009 2009;55(1):47-50.
5. Weiss JP, Schlecker BA, Wein AJ, Hanno PM. Preservation of periprostatic autonomic nerves during total perineal prostatectomy by intrafascial dissection. Urology. 1985 1985;26(2):160-3.
6. Glassberg KI, Braren V, Duckett JW, Jacobs EC, King LR, Lebowitz RL, et al. Suggested terminology for duplex systems, ectopic ureters and ureteroceles. The Journal of urology. 1984 1984;132(6):1153-4.
7. Hartman GW, Hodson CJ. The duplex kidney and related abnormalities. Clinical radiology. 1969;20(4):387-400.
8. Privett JT, Jeans WD, Roylance J. The incidence and importance of renal duplication. Clinical radiology. 1976 1976;27(4):521-30.
9. Campbell M, Harrison J. Anomalies of the Ureter. In: Campbell M, editor. Urology. 3rd ed. Philadelphia: WB Saunders; 1970.
10. Schlussel R, Retik A. Ectopic ureter, ureterocele, and other anomalies of the ureter. In: Wein A, Kavoussi L, Novick A, editors. Campbell-Walsh Urology. 9th ed. Philadelphia: Saunders Elsevier; 2008. p. 3383-422.
11. Jelloul L, Valayer J. Ureteroureteral anastomosis in the treatment of reflux associated with ureteral duplication. The Journal of urology. 1997 1997;157(5):1863-5.
12. Ellerker AG. The extravesical ectopic ureter. The British journal of surgery. 1958 1958;45(192):344-53.
13. Ejaz T, Malone PS. Male duplex urinary incontinence. The Journal of urology. 1995 1995;153(2):470-1.
14. Chacko JK, Koyle MA, Mingin GC, Furness PD, 3rd. Ipsilateral ureteroureterostomy in the surgical management of the severely dilated ureter in ureteral duplication. The Journal of urology. 2007 2007 Oct (Epub 2007 Aug;178(4 Pt 2):1689-92.
15. Costa HC, Moreira RJ, Fukunaga P, Fernandes RC, Boni RC, Matos AC. Anatomic variations in vascular and collecting systems of kidneys from deceased donors. Transplantation proceedings. 2011 2011;43(1):61-3.