Editorial: Touching the future – 3D printing facilitates preoperative planning, realistic simulation and enhanced precision in RALPN
Practice taking that match-winning penalty knowing which way the keeper will dive, or take that last putt knowing the lie of the green; it would be very handy wouldn’t it?
Virtual reality (VR), augmented reality (AR), computer-generated images (CGI), and stereotactic overlay, have all been documented as adjuncts in enhancing operative patient care through planning, simulation and increased precision. But what if you could actually handle the specimen and practice operating on a model to refine operative technique before the definitive procedure? With three-dimensional (3D) printing this has now become a reality.
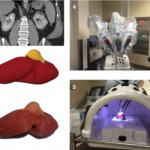
The work of von Rundstedt et al. [1] has the potential to transform surgical planning, operative accuracy, and training, with the development of a life-like kidney and tumour model. Their paper validates a patient-specific simulation protocol to assist in surgical decision-making through operative rehearsal. They assessed the benefits of 10 patient-specific 3D renal models for preoperative planning using tissue-like silicone, and performed model tumour excision with the robot before actual robot-assisted laparoscopic partial nephrectomy (RALPN). Nephrometry scores for tumours ranged from 7 to 11, with a relatively large mean maximal tumour diameter of 40.6 mm. In validating the model the investigators compared resection times between the model and patient (6.61 vs 7.93 min, P = 0.16) and tumour volumes between computer model, excised model, and excised tumour (38.88 vs 38.50 vs 41.79 mm3, P = 0.98), showing no significant differences.
The key principles in nephron-sparing surgery are adequate oncological excision, whilst preserving maximal renal parenchyma, with minimal ischaemia time, and avoiding complications. RALPN is challenging for complex tumours, with an extended learning curve, due partly to limitations in accurate surgical planning and surgical technique. Key anatomical considerations for planning including kidney orientation, tumour position and depth, and locality of adjacent anatomical structures (vessels, collecting system), are difficult to appreciate on conventional two-dimensional axial imaging platforms; with variance in imaging and model-planned approaches clearly noted in previous studies [2].
The development of nephrometry scores have been designed to predict surgical complexity along with various simulation and modelling reconstructions to aid excisional techniques. Several other surgical specialties (orthopaedics, maxillofacial and craniofacial surgery, neurosurgery, plastic surgery [3]) have used 3D-printing technology for organ/lesion modelling or to produce accurate imaging-based prostheses. In the era of minimally invasive surgery and personalised medicine, 3D printing can be a powerful tool for uro-oncologists to better understand individual tumour characteristics and anatomical variations.
Currently there is limited published data on 3D renal tumour printing. This paper [1] represents the first model and validation of its kind. Previous studies of 3D-printed kidneys with renal masses have been reported but limited to depictions of anatomical visualisation of arteries, collecting system and the tumour itself [4, 5]. This is the first time that a model not only provides a 3D representation of the tumour anatomy but also allows high-fidelity simulated excision. Construct validity of these reproductions has been assessed and demonstrate a striking similarity in tumour volume, morphology, and resection time, the main limitations of previous studies [4].
Preoperative planning has never achieved this accuracy before. Operative rehearsal significantly altered the ultimate approach to tumour excision in several cases as noted by the authors. In addition, 3D printing represents a breakthrough in surgical training as it offers a great opportunity, especially in facilities were wet laboratories are not available.
With the small sample assessed and the subjective nature of the surgical technique modification between model and tumour excision, early generalisation may not be appropriate. Other limitations include the models inability to replicate viable orthotopic anatomy such as adjacent organs, dissection planes, perinephric fat thickness/adherence, and bleeding; while also excluding the renorrhaphy component of the procedure. Additionally, production costs and 3D printer access may be an initial deterrent to widespread use of this technique; however, it does address the lack of tactile feeling in AR or VR, avoids the specialised facilities required by animal or cadaveric models, can readily be accurately reproduced, and most importantly provide an accurate anatomical representation of the individual patient.
This is an important and interesting paper as it presents and validates a novel model with extirpative technique in a prospective manner. It provides a life-like model useful for patient education, procedural practice with realistic simulation, an accurate training platform, and is the easiest to access given current technology. Clinical trials are needed to confirm how 3D modelling is ultimately useful in: i) improving patient education, ii) enhancing surgical training, and iii) conferring superior clinical outcomes. Evolution of 3D printers and shrinking production costs will eventually contribute to the widespread usage of this technology.
Further development will provide functional models that replicate not only macroscopic structures but elements such as the collecting system, segmental vessels, and bleeding parenchyma. However, it may be that high-fidelity VR simulators or CGI that can generate patient-specific graphics or even provide an intraoperative stereotactic 3D overlay to guide tumour excision may eventually supersede 3D modelling. Urology has entered the 3D printing era. This study [1] shows that 3D printing is both a feasible and useful technique that may enhance current practice, while providing an improved training platform. The future is here today.
How to Cite this Article
von Rundstedt, F.-C., Scovell, J. M., Agrawal, S., Zaneveld, J. and Link, R. E. (2017), Utility of patient-specific silicone renal models for planning and rehearsal of complex tumour resections prior to robot-assisted laparoscopic partial nephrectomy. BJU International, 119: 598–604. doi: 10.1111/bju.13712