Article of the week: A randomized trial comparing bipolar TUVP with GreenLight laser PVP for treatment of small to moderate benign prostatic obstruction
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urological community and a podcast prepared by one of our Resident podcasters; we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, we recommend this one.
A randomized trial comparing bipolar transurethral vaporization of the prostate with GreenLight laser (xps‐180watt) photoselective vaporization of the prostate for treatment of small to moderate benign prostatic obstruction: outcomes after 2 years
Fady K. Ghobrial, Ahmed Shoma, Ahmed M. Elshal, Mahmoud Laymon, Nasr El-Tabey, Adel Nabeeh and Ahmed A. Shokeir
Urology Department, Urology and Nephrology Center, Mansoura University, Mansoura, Egypt
Abstract
Objective
To test the non‐inferiority of bipolar transurethral vaporization of the prostate (TUVP) compared to GreenLight laser (GL) photoselective vaporization of the prostate (PVP) for reduction of benign prostatic hyperplasia‐related lower urinary tract symptoms in a randomized trial.
Methods
Eligible patients with prostate volumes of 30–80 mL were randomly allocated to GL‐PVP (n = 58) or bipolar TUVP (n = 61). Non‐inferiority of symptom score (International Prostate Symptom Score [IPSS]) at 24 months was evaluated. All peri‐operative variables were recorded and compared. Urinary (IPSS, maximum urinary flow rate and post‐void residual urine volume) and sexual (International Index of Erectile Function‐15) outcome measures were evaluated at 1, 4, 12 and 24 months. Need for retreatment and complications, change in PSA level and health resources‐related costs of both procedures were recorded and compared.
Results
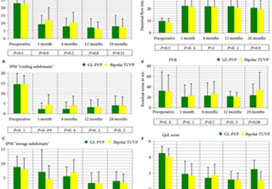
Baseline and peri‐operative variables were similar in the two groups. At 1, 4, 12 and 24 months, 117, 116, 99 and 96 patients, respectively, were evaluable. Regarding urinary outcome measures, there was no significant difference between the groups. The mean ± sd IPSS at 1 and 2 years was 7.1 ± 3 and 7.9 ± 2.9 (P = 0.8), respectively, after GL‐PVP and 6.3 ± 3.1 and 7.2 ± 2.8, respectively, after bipolar TUVP (P = 0.31). At 24 months, the mean difference in IPSS was 0.7 (95% confidence interval −0.6 to 2.3; P = 0.6). The median (range) postoperative PSA reduction was 64.7 (25–99)% and 65.9 (50–99)% (P = 0.006) after GL‐PVP, and 32.1 (28.6–89.7)% and 39.3 (68.8–90.5)% (P = 0.005) after bipolar TUVP, at 1 and 2 years, respectively. After 2 years, retreatment for recurrent bladder outlet obstruction was reported in eight (13.8%) and 10 (16.4%) patients in the GL‐PVP and bipolar TUVP groups, respectively (P = 0.8). The mean estimated cost per bipolar TUVP procedure was significantly lower than per GL‐PVP procedure after 24 months (P = 0.01).
Conclusions
In terms of symptom control, bipolar TUVP was not inferior to GL‐PVP at 2 years. Durability of the outcome needs to be tracked. The greater cost of GL‐PVP compared with bipolar TUVP is an important concern.