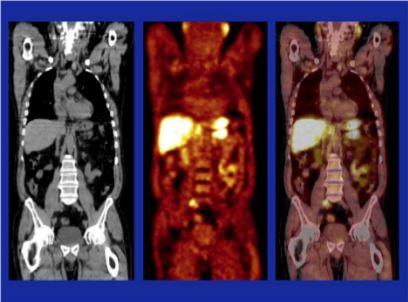
Article of the Week: 11C-acetate PET/CT imaging for detection of recurrent disease after RP or RT in patients with PCa
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
11C-acetate positron-emission tomography/computed tomography imaging for detection of recurrent disease after radical prostatectomy or radiotherapy in patients with prostate cancer
Abstract
Objectives
To evaluate, in a prospective study, the effectiveness of computed tomography (CT)-matched 11C-acetate (AC) positron-emission tomography (PET) in patients with prostate cancer (PCa) who had prostate-specific antigen (PSA) relapse after radical prostatectomy (RP) or radiotherapy (RT).
Patients and Methods
In 103 relapsing patients after RP (n = 97) or RT (n = 6) AC-PET images and CT scans were obtained. In patients with AC-PET-positive results with localized PCa recurrence, detected lesions were resected and histologically verified or, after local RT, followed-up by PSA testing. Patients with distant disease on AC-PET were treated with androgen deprivation/chemotherapy.
Results
Of 103 patients, 42 were AC-PET-positive. PSA levels were <1.0, <2.0 and <4.0 ng/mL in six, 16 and 20 patients, respectively. In 25/42 patients AC-PET suggested lymph node metastases: 16/25 patients underwent surgery (10/16 metastasis, 6/16 inflammation); 9/25 patients underwent RT of lymph node metastases, which was followed by decreasing PSA level. In 17/42 patients who had distant disease, systemic treatment was commenced. Combining patients who underwent surgery and those who underwent RT, 19/25 patients were true-positive in terms of AC-PET (positive predictive value 76%). In 5/19 patients, PSA level was <2.0 ng/mL, in 2/19 patients it was <1.0 ng/mL and in 14/19 patients it was 5.4–23.1 ng/mL. In AC-PET-positive patients after surgery or RT (without androgen deprivation), median (range) time to renewed PSA increase was 6 (5–9) months.
Conclusions
Only a minority of patients with relapsing PCa appear to benefit from AC-PET for guiding potential local treatment. False-positive results show that factors other than tumour metabolism induce increased AC uptake. The time free of recurrence after local treatment was shorter than expected.