Editorial: Well‐being beyond the bladder. How do we improve the overall health of patients with bladder cancer?
Declines in quality of life and physical function are commonly associated with all cancers 1, and in this month’s issue of BJUI, Smith et al. 2 describe the changes in quality of life that occur specifically in patients with bladder cancer. The authors examine 535 individuals with bladder cancer (of whom 77 [14%] had invasive disease) and matched them to 2770 non‐cancer controls using propensity scores. The Surveillance, Epidemiology and End Results (SEER) registry was linked with the Medicare Health Outcomes Survey. This dataset represents linkages of population‐based SEER data with survey data for Medicare‐managed enrollees. In this study, patients were surveyed at different times with respect to their diagnosis and the authors identified all patients who were surveyed some time before and after their diagnosis. By harnessing this dataset, the authors describe changes that occur in mental and physical function. The authors should be commended for conducting an analysis that seeks to quantify the impact of a bladder cancer diagnosis on multiple dimensions affecting quality of life.
A few findings are worth highlighting. First, the quality of life of a patient with bladder cancer declines more between a pre‐diagnosis and post‐diagnosis assessment as compared with matched, non‐cancer controls. As any urological oncologist can attest, a bladder cancer diagnosis causes permanent changes to a patient’s life. Second, people with bladder cancer have deficits in multiple domains of well‐being and not just in physical function. Third, people with bladder cancer have impairments in well‐being whether they have non‐invasive or invasive disease. Fourth, decrements were more pronounced in those with invasive disease. In fact, patients who underwent cystectomy had statistically significant declines in nearly all physical domains and similar declines in mental health‐related quality of life across several domains, including emotional, vitality and social functioning. Lastly, a predictor of a significant decrease in both the physical component and mental component score included a diagnosis of recent depression. This insightful study shows the potential impact of a bladder cancer diagnosis on mental and physical health‐related quality of life.
So, with these detriments in mind, can urologists do anything to address these declines in quality of life for patients with bladder cancer?
In clinical practice, urologists may be able to play an active role in mitigating the negative consequences of therapy, be it for invasive or non‐invasive disease. If a urologist is following a patient with non‐muscle‐invasive bladder cancer, then there are clinical visits for cystoscopy, intravesical instillation and follow‐up, during which a provider can regularly check in with a patient and offer recommendations. If a patient has muscle‐invasive bladder cancer, they are typically seen a few times before surgery and there is an incentive to address potentially modifiable sources of morbidity before a major operation plagued by complications.
While encouraging healthy behaviours is common sense and may help some patients, understanding the difference between motivating self‐care (e.g. coaching our patients) and recommending programmes that are scientifically established and effective (e.g. recommending a programme proven in a randomized controlled trial) are different. One major challenge in promoting healthy behaviours in our patients is understanding their mindset, i.e. their motivation to make meaningful change. The Transtheoretical Model is a biopsychosocial model that conceptualizes intent for changing behaviour: pre‐contemplation, contemplation, preparation, action, maintenance and termination 3. Based on a continuum of patient activation and knowledge of these stages, interventions can be designed more effectively and focused on individuals. Conversation content, clinician effort and clinical resources can be judiciously allotted instead of offering all options to all patients.
The presence of validated interventions that have been determined to consistently improve quality of life is evolving. A new area of preoperative care known as prehabilitation, is being studied in patients with cancer and seeks to optimize preoperative factors, such as increasing fitness, improving nutritional status, encouraging smoking cessation and decreasing anxiety 4. Although studies vary in quality, content and outcomes measured 5, there is still an opportunity to exercise common sense and make practical suggestions.
For busy urologists who manage patients with bladder cancer, any patient can benefit from:
- Mindful conversations: having open and regular communication about quality of life.
- Measurements: tracking patient‐reported outcome measures longitudinally to follow well‐being systematically and identify detrimental changes early.
- Multidisciplinary resources: offer support (Fig. 1) based on conversations (#1) and scores (#2).
Conversations only require a little provider time, monitoring patient‐reported outcomes can be facilitated by the use of technology such as the electronic health record, and most institutions have previously established resources that patients can use during their care. Strategies may be low‐cost, quick and capable of helping patients or caregivers. Also, data show that routine assessment of patient‐reported outcome measures in patients with advanced cancers may be associated with improved overall survival 6.
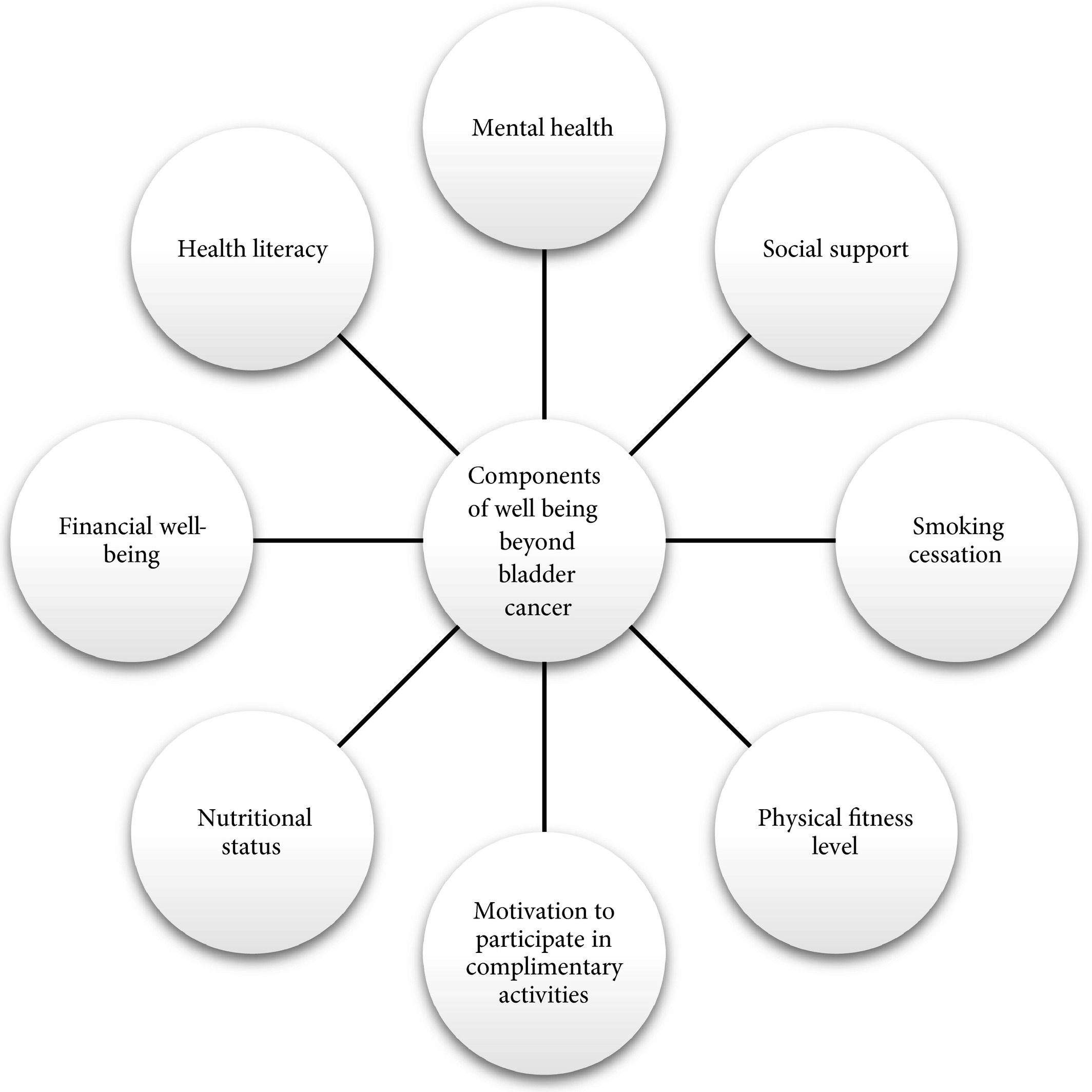
Figure 1
Acknowledging that other dimensions of health are affected after a bladder cancer diagnosis may allow us to track, address and ultimately improve the health of our patients. When we care for patients with bladder cancer, focusing cancer treatment is paramount; however, we can also extend this treatment by being cognisant of quality of life. Complementing oncological care with efforts to promote health in other ways allows us to promote well‐being and treat these patients beyond the bladder.
- 1 Petrick JL, Reeve BB, Kucharska‐Newton AM et al. Functional status declines among cancer survivors: trajectory and contributing factors. Journal of Geriatric Oncology 2014; 5: 359–67
- 2 Smith AB, Jaeger B, Pinheiro LC et al. Impact of bladder cancer on health‐related quality of life. BJU Int 2018; 121: 549–57
- 3 Prochaska JO, Velicer WF. The transtheoretical model of health behavior change. Am J Health Promot 1997; 12: 38–48
- 4 Silver JK, Baima J. Cancer prehabilitation: an opportunity to decrease treatment‐related morbidity, increase cancer treatment options, and improve physical and psychological health outcomes. Am J Phys Med Rehabil 2013; 92: 715–27
- 5 Mayo NE, Feldman L, Scott S et al. Impact of preoperative change in physical function on postoperative recovery: argument supporting prehabilitation for colorectal surgery. Surgery 2011; 150: 505–14. https://doi.org/10.1016/j.surg.2011.07.045
- 6 Basch E, Deal AM, Dueck AC et al. Overall survival results of a trial assessing patient‐reported outcomes for symptom monitoring during routine cancer treatment. JAMA 2017; 318: 197–8