Editorial: Should we care more about SPARE?
Huddart et al. [1] report the results of a phase III clinical trial (SPARE) evaluating the feasibility of randomising participants with cT2/T3 muscle-invasive bladder cancer (MIBC) to either radiation or radical cystectomy (RC) following neoadjuvant chemotherapy. Whilst attempting to address an important, in fact crucial ongoing point of debate, due to poor patient accrual (45 participants recruited in 30 months) the study was terminated early. Additionally, compliance with study assignment was low, with as many as 24% of participants electing to proceed with a treatment arm that was the opposite of what they were randomised to. This underscores the problems with obtaining randomised data in an era where patient and clinician preference drive clinical decision-making.
Whilst well-performed prospective studies show acceptable results with bladder-sparing approaches (69% complete response and 71% 5-year disease-specific survival [2]), no randomised clinical trials comparing bladder sparing with RC have demonstrated equivocal results. As non-randomised studies are subject to selection bias [many patients with large bulky T3 tumours or those with carcinoma in situ (CIS) or hydronephrosis have not met inclusion criteria for trials of bladder sparing], it is often debated as to whether bladder sparing is appropriate for the entire population of patients with MIBC or a selected subset. This is especially important in a deadly disease such as bladder cancer, where we often have only one chance to get it right and hence recent guidelines state that bladder sparing and radical options should be discussed with the patient.
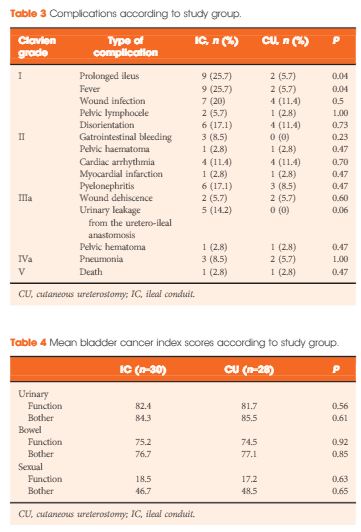
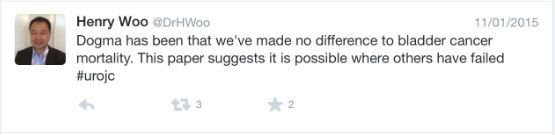
Whilst we acknowledge the limitations based on the number of participants analysed, these data show some interesting trends [1]. There appears to be more local recurrence with radiation therapy (69%) compared to RC (15%), this despite confirmation of ≤cT1 disease after neoadjuvant chemotherapy. And while most of the local failures are attributed to non-muscle-invasive recurrences; additional treatments, patient anxiety, and potential salvage RC, as well as the cost of surveillance, must be considered in reflecting on these results. It is unclear whether these factors are outweighed by the perceived lower toxicity in these patients.
It is our opinion that until randomised studies show equivalency, radiation-based approaches should definitely be discussed with all patients but patients should also be guided as to who are ideal candidates. Ideal candidates are those who have non-variant histology (pure urothelial carcinoma), non-bulky (minimal) invasive T2 cancer, absence of CIS, absence of a three-dimensional mass on imaging or examination, absence of hydronephrosis, and have an adequate bladder capacity [3]. The role of multidisciplinary care is paramount, maximal transurethral resection is a critical initial step, as incomplete resections can potentially double the odds of eventual RC in bladder-sparing protocols [4]. Additionally, concomitant chemotherapy has shown improved survival and should be considered standard of care based on a randomised control trial [5]. The addition of neoadjuvant chemotherapy is unknown and needs to be further evaluated. In addition to the treatment itself, it is clear that vigilant surveillance is critical in identifying patients at highest risk of failure and requires a combination of both cystoscopy and imaging, with expedient salvage RC.
Beyond the challenges of treating patients with MIBC, this report from Huddart et al. [1] reflects the larger issue of clinical trial accrual. As mentioned, patients and clinicians often have predetermined notions about the ‘best’ course of action, even in the context of a randomised clinical trial. These limitations have plagued early closure of other trials in bladder cancer as well. Similarly in prostate cancer, comparative treatment trials of radiation and surgery are limited as patients are reluctant to relinquish decisions about their treatment. Clearly, an intensive effort is necessary to create clinical trials that are palatable to a multi-disciplinary treatment team committed to answering tough therapy questions. MIBC is no different, where we often offer differing treatment modalities without having quality comparative data, which is a disservice to our patients who look to us to guide their treatment approaches based on best available hard evidence. We again commend the authors for their well-designed clinical trial and presenting their results and challenges from the SPARE trial.
References