Article of the Week: DSNB for Penile Cancer
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Dynamic sentinel lymph node biopsy for penile cancer: a comparison between 1- and 2-day protocols
Objective
To determine the outcome of clinically negative node (cN0) patients with penile cancer undergoing dynamic sentinel node biopsy (DSNB), comparing the results of a 1- and 2-day protocol that can be used as a minimal invasive procedure for staging of penile cancer.
Patients and Methods
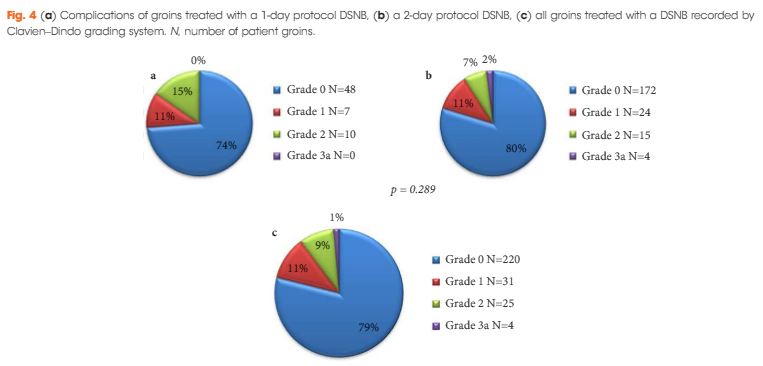
This is a retrospective analysis of 151 cN0 patients who underwent DSNB from 2008 to 2013 for newly diagnosed penile cancer. Data were analysed per groin and separated into groups according to the protocol followed. The comparison of the two protocols involved the number of nodes excised, γ-counts, false-negative rates (FNR), and complication rates (Clavien–Dindo grading system).
Results
In all, 280 groins from 151 patients underwent DSNB after a negative ultrasound ± fine-needle aspiration cytology. The 1-day protocol was performed in 65 groins and the 2-day protocol in 215. Statistically significantly more nodes were harvested with the 1-day protocol (1.92/groin) compared with the 2-day protocol (1.60/groin). The FNRs were 0%, 6.8% and 5.1%, for the 1-day protocol, 2-day protocol, and overall, respectively. Morbidity of the DSNB was 21.4% for all groins, and 26.2% and 20.1% for the 1-day and 2-day protocols, respectively. Most of the complications were of Clavien–Dindo Grade 1–2.
Conclusions
DSNB is safe for staging patients with penile cancer. There is a trend towards a 1-day protocol having a lower FNR than a 2-day protocol, albeit at the expense of a slightly higher complication rate.