Editorial: Low rates of bone density testing in prostate cancer survivors on androgen‐deprivation therapy: where do we go from here?
In this month’s issue of the BJU International, Kirk et al. 1 describe their findings regarding an important issue in the care of prostate cancer survivors on androgen‐deprivation therapy (ADT): the underuse of bone density testing (BDT) to screen for osteoporosis. ADT is the commonest systemic therapy in patients with prostate cancer, used in both metastatic and localised settings. Whilst it has clear survival benefits, ADT is also associated with harms including cardiovascular, cognitive, and metabolic side‐effects, as well as an increased risk of osteoporosis and fractures. These bone‐related complications are costly from a quality‐of‐life and financial perspective, especially given the critical importance of mobility in maintaining performance status and cardiovascular health during cancer treatment 2, 3. Consequently, most clinical practice guidelines include osteoporosis screening as a recommendation for men undergoing ADT.
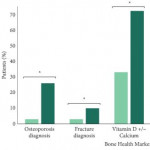
In their study, ‘The implications of baseline bone health assessment at initiation of androgen‐deprivation therapy for prostate cancer’, the authors describe patterns of use of BDT and diagnosis of osteoporosis amongst men treated for prostate cancer in the USA Veterans Affairs (VA) system within a 3‐year period following ADT initiation. There was a statistically significant increase in the BDT rate throughout the study period; however, overall BDT remains uncommon amongst patients with prostate cancer on ADT, used in only 15% of men in their cohort. Unsurprisingly, patients who received BDT were more likely to be diagnosed with osteoporosis, be diagnosed with a fracture, and receive treatment with vitamin D, calcium and bisphosphonates. The authors acknowledge an important limitation about the applicability of their VA study to the civilian health population. However, given that the VA and military health systems perform as well, if not better, on several important metrics in prostate cancer care 4, 5, these results should not be ignored simply because they were obtained in the military health system.
The increase in BDT screening throughout the study may be attributable to increased awareness of guidelines published during the study period. However, the overall BDT rate remains low. This may be explained by insufficient access, lack of information technology, as well as more nebulous aspects of care such as physician culture, beliefs, and habits 6.
Studies such as this are vital to identify opportunities for improving care delivery. What are needed next are innovations to optimise the delivery of care for patients treated with ADT. Whilst improving BDT adherence may lack the cachet of next‐generation targeted therapies, this is an example of the kind of simple, measurable area where improvement in care delivery systems may yield large benefits.
There are many possible avenues for success: quality improvement collaboratives are one well‐known innovation, which may be applicable to this area: examples, such as the Michigan Urological Surgery Improvement Collaborative (MUSIC) and the AUA Quality Registry (AQUA) are success stories, but to our knowledge there are no published studies specifically attempting to improve adherence to BDT guidelines within these cohorts. Other practice‐based innovations include navigators and multidisciplinary cancer teams, either of which may yield improvements in guideline adherence. Online patient support groups can raise awareness. And although we all know how electronic reminders have frustrated countless physicians, electronic reminders about recommended tests and interventions may be an important tool. At our institution, a Prostate Cancer Foundation grant is funding the development of a mobile health app, which is targeted exclusively at men receiving ADT for prostate cancer. This app will encourage physical activity and healthy eating, which can both support bone health.
In our view, the issue of bone screening is a clear example of where innovative strategies to improve care delivery and guideline adherence may make a big difference for men living with prostate cancer. We look forward to seeing more in the years to come.
- 1 Kirk PS, Borza T, Shahinian VB et al. The implications of baseline bone‐health assessment at initiation of androgen‐deprivation therapy for prostate cancer. BJU Int2018; 121: 558–64
- 2 Sharifi N, Gulley JL, Dahut WL. Androgen deprivation therapy for prostate cancer. JAMA 2005; 294: 238–44
- 3 Shahinian VB, Kuo YF, Freeman JL, Goodwin JS. Risk of fracture after androgen deprivation for prostate cancer. N Engl J Med 2005; 352: 154–64
- 4 Cole AP, Jiang W, Lipsitz SR et al. The use of prostate specific antigen screening in purchased versus direct care settings: data from the TRICARE® military database. J Urol 2017; 198: 1295–300
- 5 Cullen J, Brassell SA, Chen Y et al. Racial/Ethnic patterns in prostate cancer outcomes in an active surveillance cohort. Prostate Cancer 2011; 2011: 234519
-
- 6 Netwrok for Excellence in Health Innovation (NEHI). Improving Physician Adherence to Clinical Practice Guidelines ‐ Barriers and strategies for change. Available at: https://www.nehi.net/publications/53-improving-physician-adherence-to-clinical-practice-guidelines/view. Accessed January 2018 Google Scholar