Editorial: How active should active surveillance be?
Many investigators, including those from Johns Hopkins University (JHU) and the Prostate cancer Research International: Active Surveillance project (PRIAS), have provided meaningful data to strongly support the increasing use of active surveillance (AS) across the world. There are a multitude of strategies to minimise excessive rates of prostate cancer over detection and overtreatment. After the diagnosis of prostate cancer, the single best is AS for appropriately selected men.
For decades, the concept of not treating a prostate cancer in otherwise healthy men, even if low-grade and low-volume was typically considered nihilistic and heretical, particularly in the USA. Thankfully, data have largely made this line of thinking anachronistic. The era of sensibly applied AS is upon us, and single-institution series with intermediate-term follow-up are excellent, with exceedingly low rates of metastasis or cancer-related death. However, we await longer-term (>10 years) outcomes from the contemporary PSA screening era.
The ‘success’ of AS is largely dependent on the entry criteria, follow-up strategies, and indications for curative intervention. Highly restrictive inclusion criteria, rigorous biopsy based follow-up and strict definitions of reclassification triggering treatment have produced superb outcomes. Critics appropriately argue these criteria exclude a significant proportion of men with a low rate of requiring treatment or having metastases, if allowed on AS. Conversely, other programmes with looser entry criteria, more lax follow-up, and relaxed indications for intervention will be more inclusive and have lower rates of immediate or delayed intervention but must be counterbalanced against the expected higher rate of metastases or death.
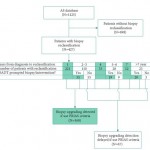
The current study [1] evaluates two different AS follow-up strategies from JHU and PRIAS. In general, JHU uses annual biopsies with progression defined as a new PSA density >0.15 ng/mL/mL or increasing tumour volume or grade beyond a certain threshold, while PRIAS recommends less frequent biopsies (years 1, 4, and 7) while relying on serological (PSA doubling time, PSADT) alongside histological indicators for defining progression and recommending treatment.
Not surprisingly, different strategies lead to varying expected outcomes. Among the JHU patients, 38% were reclassified at a median of 2.1 years. Nearly two-thirds of the reclassified would have been identified at the PRIAS year 1 or triennial biopsies with 16% identified between PRIAS biopsies at a median delay of 1.9 years. The unanswerable but incredibly important question is whether this delay is essentially a non-issue, perhaps a favourable attribute (more AS time without compromising cure rates), or clinically disastrous (patients no longer curable).
PRIAS relies heavily on PSA kinetics, which can be a double-edged sword. Among men in the JHU programme with >5 years follow-up, 11% would have delayed reclassification compared with PRIAS at a median time of 4.7 years. Additionally, 12% would have undergone intervention due to PRIAS-defined PSADT but not progressed based on the JHU protocol. It is convenient and perhaps intuitive that PSA kinetics should predict progression and meaningful clinical events for men on AS; however, the data from multiple studies have simply not supported this concept [2, 3].
The JHU programme has restrictive entry rules compared with most other programmes, a rigorous biopsy based follow-up protocol, and strict criteria to treat, which is exactly why no metastasis or death have been reported among 769 men, some with up to 15 years follow-up[4]. Guidelines are needed but should not be overly prescriptive or rigid. For example, a surveillance biopsy showing a single core of Gleason 6 encompassing 60% of the total core or three cores of Gleason 6 with total cancer length of 3 mm would lead to a recommendation of treatment according to published JHU criteria. Many of us would not be phased with these biopsy reports and comfortably recommend ongoing AS.
Data from AS series are very encouraging but it is highly likely we can do even better. For example, 10-year cancer-specific survival is 97% in the Sunnybrook AS experience and all five cancer-related deaths occurred in patients that would not meet most contemporary AS entry criteria [5, 6]. I am hopeful and confident that emerging data incorporating MRI imaging, serum biomarkers (e.g. prostate health index), or tissue-based biomarkers (e.g. Prolaris, Oncotype Dx) will provide us with a more comprehensive understanding of these men’s cancer such that tailored, evidence-based recommendations can be even more accurate.
There is much yet to be learned about AS and this study [1] adds to our knowledge. Surveillance for prostate cancer is definitely active, but it is also dynamic and evolving.



That’s a sensible response, thanks Scott. I think this paper is essentially historical with the relatively recent adoption of Transperineal Biopsy & MpMRI. Are there any of us that solely rely on TRUS biopsy for diagnosing prostate cancer still? A more important study would be how many clinically significant cancers slip through the contemporary diagnostic net, and ultimately (as you allude to) does this change any outcomes for those few men?