Article of the Week: Profiling microRNA from nephrectomy and biopsy specimens
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Profiling microRNA from nephrectomy and biopsy specimens: predictors of progression and survival in clear cell renal cell carcinoma
Abstract
Objective
To identify microRNA (miRNA) characteristic of metastatic clear cell renal cell carcinoma (ccRCC) and those indicative of cancer-specific survival (CSS) in nephrectomy and biopsy specimens. We also sought to determine if a miRNA panel could differentiate benign from ccRCC tissue.
Materials and Methods
RNA was isolated from nephrectomy and kidney biopsy specimens (n = 156 and n = 46, respectively). Samples were grouped: benign, non-progressive, and progressive ccRCC. MiRNAs were profiled by microarray and validated by quantitative reverse transcription-polymerase chain reaction. Biomarker signatures were developed to predict cancer status in nephrectomy and biopsy specimens. CSS was examined using Kaplan–Meier and Cox proportional hazards analyses.
Results
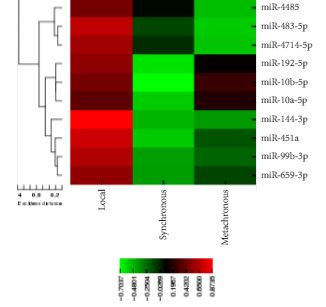
Microarray analysis revealed 20 differentially expressed miRNAs comparing non-progressive with progressive tumours. A biomarker signature validated in nephrectomy specimens had a sensitivity of 86.7% and a specificity of 92.9% for differentiating benign and ccRCC specimens. A second signature differentiated non-progressive vs progressive ccRCC with a sensitivity of 93.8% and a specificity of 83.3%. These biomarkers also discriminated cancer status in biopsy specimens. Levels of miR-10a-5p, -10b-5p, and -223-3p were associated with CSS.
Conclusion
This study identified miRNAs differentially expressed in ccRCC samples; as well as those correlating with CSS. Biomarkers identified in this study have the potential to identify patients who are likely to have progressive ccRCC, and although preliminary, these results may aid in differentiating aggressive and indolent ccRCC based on biopsy specimens.