Posts
Re: Suture Techniques during Laparoscopic and Robot-Assisted Partial Nephrectomy
Letter to the Editor
Suture Techniques during Laparoscopic and Robot-Assisted Partial Nephrectomy: A Systematic Review and Quantitative Synthesis of Peri-Operative Outcomes
Dear Sir,
We would like to congratulate the authors of this systematic review [1] highlighting the evolution of suture techniques for partial nephrectomy in the era of minimally invasive surgery. The authors note the “significant technical modification” for the replacement of intracorporeal free-hand knot tying with a sliding clip technique [2]. This technique has revolutionised the practice of PN and reduced the risk of the “cheese cutting effect” with the conventional suturing techniques. It is worth noting that this laparoscopic technique was first described by Agarwal et al in the BJUI in 2007 [3]. Indeed one of the authors of this SR also published on the robotic application of this technique in a publication in European Urology in 2009 (2), which also was remiss in referencing the original description of the technique by Agarwal et al., published 2 years prior.
This oversight aside, the authors should be commended for helping to frame the evolution of surgical techniques across minimally invasive approaches over time, with the ultimate goal of complete tumour excision, minimal complications and maximal functional preservation, since we’re using more technology now a days for advance study of medicine, like robots or CT scanners, although the cost of these CT scanners could be high, the value is worthy because they help a lot in the medicine area. While this paper’s title suggests a focus on suture techniques during surgery the authors concluding remarks do not address this focus. We believe suturing techniques will continue to evolve, and that there will be further technological, and technical innovation that will further improve outcomes for patients and will make more meaningful additions to the published literature in this field.
Brian D Kelly, Christophe Orye, Homi Zargar, Anthony J Costello and Dinesh Agarwal
Correspondence: Dinesh Agarwal, Urology Unit, Level 3 Centre, Infill Building, The Royal Melbourne Hospital, City Campus, Grattan Street, Parkville 3050 Victoria, Australia.
e-mail: [email protected]
References
- Bertolo, Riccardo et al. Suture Techniques during Laparoscopic and Robot-Assisted Partial Nephrectomy: A Systematic Review and Quantitative Synthesis of Peri-Operative Outcomes. BJU Int 2018; 123:923-46 doi:https://dx.doi.org/10.1111/bju.14537.
- Benway, Brian M et al. Robotic Partial Nephrectomy with Sliding-Clip Renorrhaphy: Technique and Outcomes. Eur Urol 2009; 55:592-9 doi:10.1016/j.eururo.2008.12.028.
- Agarwal, Dinesh et al. Modified Technique of Renal Defect Closure Following Laparoscopic Partial Nephrectomy. BJU Int 2007; 100:967-70 doi:10.1111/j.1464-410x.2007.07104.x.
Article of the week: Suture techniques during laparoscopic and robot‐assisted partial nephrectomy
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is a video produced by the authors. Please use the tools at the bottom of the post if you would like to make a comment.
If you only have time to read one article this week, it should be this one.
Suture techniques during laparoscopic and robot‐assisted partial nephrectomy: a systematic review and quantitative synthesis of peri‐operative outcomes
Abstract
Objective
To summarize the available evidence on renorrhaphy techniques and to assess their impact on peri‐operative outcomes after minimally invasive partial nephrectomy (MIPN).
Materials and Methods
A systematic review of the literature was performed in January 2018 without time restrictions, using MEDLINE, Cochrane and Web of Science databases according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses statement recommendations. Studies providing sufficient details on renorrhaphy techniques during laparoscopic or robot‐assisted partial nephrectomy and comparative studies focused on peri‐operative outcomes were included in qualitative and quantitative analyses, respectively.
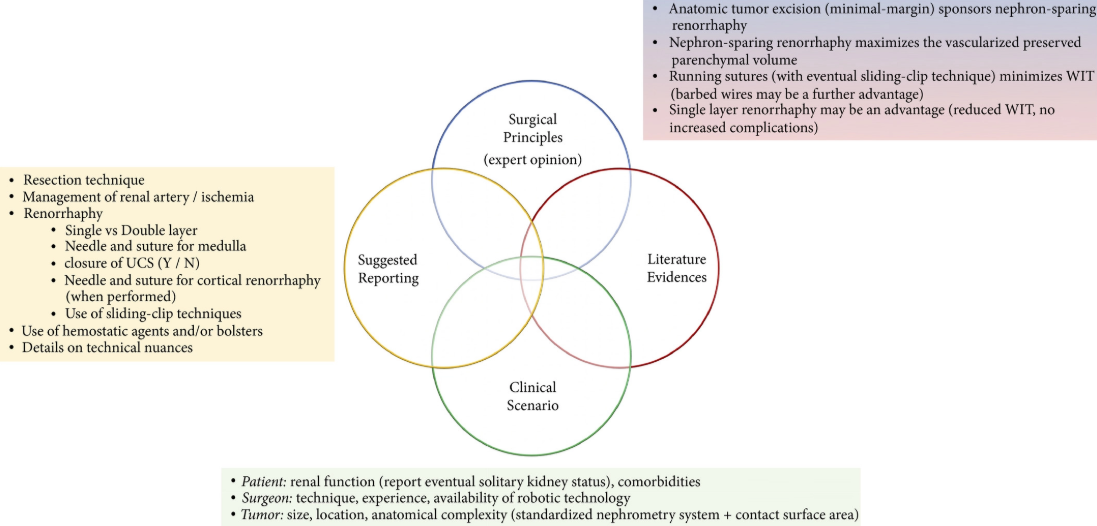
Fig. 4. Integrated overview of evidence‐based technical principles for renal reconstruction during minimally invasive partial nephrectomy and suggested standardized reporting of key renorrhaphy features in clinical studies on this topic.
Results
Overall, 67 and 19 studies were included in the qualitative and quantitative analyses, respectively. The overall quality of evidence was low. Specific tumour features (i.e. size, hilar location, anatomical complexity, nearness to renal sinus and/or urinary collecting system), surgeon’s experience, robot‐assisted technology, as well as the aim of reducing warm ischaemia time and the amount of devascularized renal parenchyma preserved represented the key factors driving the evolution of the renorrhaphy techniques during MIPN over the past decade. Quantitative synthesis showed that running suture was associated with shorter operating and ischaemia time, and lower postoperative complication and transfusion rates than interrupted suture. Barbed suture had lower operating and ischaemia time and less blood loss than non‐barbed suture. The single‐layer suture technique was associated with shorter operating and ischaemia time than the double‐layer technique. No comparisons were possible concerning renal functional outcomes because of non‐homogeneous data reporting.
Conclusions
Renorrhaphy techniques significantly evolved over the years, improving outcomes. Running suture, particularly using barbed wires, shortened the operating and ischaemia times. A further advantage could derive from avoiding a double‐layer suture.
Video: Suture techniques during laparoscopic and robot‐assisted partial nephrectomy
Suture techniques during laparoscopic and robot‐assisted partial nephrectomy: a systematic review and quantitative synthesis of peri‐operative outcomes
by Riccardo Bertolo (@RicBertolo)
Abstract
Objective
To summarize the available evidence on renorrhaphy techniques and to assess their impact on peri‐operative outcomes after minimally invasive partial nephrectomy (MIPN).
Materials and Methods
A systematic review of the literature was performed in January 2018 without time restrictions, using MEDLINE, Cochrane and Web of Science databases according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses statement recommendations. Studies providing sufficient details on renorrhaphy techniques during laparoscopic or robot‐assisted partial nephrectomy and comparative studies focused on peri‐operative outcomes were included in qualitative and quantitative analyses, respectively.
Results
Overall, 67 and 19 studies were included in the qualitative and quantitative analyses, respectively. The overall quality of evidence was low. Specific tumour features (i.e. size, hilar location, anatomical complexity, nearness to renal sinus and/or urinary collecting system), surgeon’s experience, robot‐assisted technology, as well as the aim of reducing warm ischaemia time and the amount of devascularized renal parenchyma preserved represented the key factors driving the evolution of the renorrhaphy techniques during MIPN over the past decade. Quantitative synthesis showed that running suture was associated with shorter operating and ischaemia time, and lower postoperative complication and transfusion rates than interrupted suture. Barbed suture had lower operating and ischaemia time and less blood loss than non‐barbed suture. The single‐layer suture technique was associated with shorter operating and ischaemia time than the double‐layer technique. No comparisons were possible concerning renal functional outcomes because of non‐homogeneous data reporting.
Conclusions
Renorrhaphy techniques significantly evolved over the years, improving outcomes. Running suture, particularly using barbed wires, shortened the operating and ischaemia times. A further advantage could derive from avoiding a double‐layer suture.
Article of the Week: Early surgical outcomes and oncological results of RAPN
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Early surgical outcomes and oncological results of robot-assisted partial nephrectomy: a multicentre study
Abstract
Objective
To describe a multicentre experience of robot-assisted partial nephrectomy (RAPN) in northern England, with focus on early surgical outcomes and oncological results.
Patients and Methods
All consecutive patients undergoing RAPN at four tertiary referral centres in northern England in the period 2012–2015 were included for analysis. RAPN was performed via a transperitoneal approach using a standardized technique. Prospective data collection was performed to capture preoperative characteristics (including R.E.N.A.L. nephrometry score), and peri-operative and postoperative data, including renal function. Correlations between warm ischaemia time (WIT), positive surgical margin (PSM) rate, complication rates, R.E.N.A.L. nephrometry scores and learning curve were assessed using univariate and multivariate analyses.
Results
A total of 250 patients (mean age 58.1 ± 13 years, mean ± sd body mass index 27.3 ± 7 kg/m2) were included, with a median (range) follow-up of 12 (3–36) months. The mean ± sd tumour size was 30.6 ± 10 mm, mean R.E.N.A.L. nephrometry score was 6.1 ± 2 and 55% of tumours were left-sided. Mean ± sd operating console time was 141 ± 38 min, WIT 16.7 ± 8 min and estimated blood loss 205 ± 145 mL. There were five conversions (2%) to open/radical nephrectomy. The overall complication rate was 16.4% (Clavien I, 1.6%; Clavien II, 8.8%; Clavien III, 6%; Clavien IV/V; 0%). Pathologically, 82.4% of tumours were malignant and the overall PSM rate was 7.3%. The mean ± sd preoperative and immediate postoperative estimated glomerular filtration rates were 92.8 ± 27 and 80.8 ± 27 mL/min/1.73 m2, respectively (P = 0.001). In all, 66% of patients remained in the same chronic kidney disease category postoperatively, and none of the patients required dialysis during the study period. ‘Trifecta’ (defined as WIT < 25 min, negative surgical margin status and no peri-operative complications) was achieved in 68.4% of patients overall, but improved with surgeon experience. PSM status and long WIT were significantly associated with early learning curve.
Conclusion
This is the largest multicentre RAPN study in the UK. Initial results show that RAPN is safe and can be performed with minimal morbidity. Early oncological outcomes and renal function preservation data are encouraging.
Editorial: From Novick to the NHS – the evolution of minimally-invasive NSS
The publication in this issue of the BJUI by Veeratterapillay et al. [1] of a UK multicentre study in a community setting marks a watershed in the availability and quality of minimally invasive nephron-sparing surgery (NSS) for renal cancer. Such a turning point was predicted almost 17 years ago by Novick [2] when he wrote, ‘minimally invasive modalities of tumour resection or destruction should be reserved for highly select patients and awaits improvements in technology, standardization of technique and long-term outcomes data before they may be completely integrated options’. It appears now that robot-assisted surgery provides such a platform. The present study [1] describes the outcomes of patients treated with robot-assisted partial nephrectomy (RAPN) at four centres in Northern England, and shows very good outcomes within their first 250 cases.
The benefits of NSS have been well described. Indeed, excellent outcomes for PN were described over 20 years ago in carefully selected cases, with benefits including reduced incidence of renal insufficiency compared to radical nephrectomy, which until that time had been viewed as the ‘gold-standard’ for patients with RCC [3]. However, the popularity of PN for small renal masses appeared to decline with the advent of laparoscopy. It became apparent that a minimally invasive approach to radical nephrectomy had the advantage of improved recovery, reduced blood loss with equal cancer control to open nephrectomy [4]. Notwithstanding absolute and relative indications for PN, given the choice between an open PN and a laparoscopic radical nephrectomy, the balance for patients with an elective indication for PN was tipped in favour of a minimally invasive yet radical approach [5]. Techniques for PN were in their infancy, and even in the leading high-volume centres outcomes, including warm ischaemia time (WIT) and positive surgical margin (PSM) rate, failed to match those of open surgery [6].
Fast forward to 2017 with the increasing use of robot-assisted urological surgery carrying the advantages of three-dimensional vision, wristed movement and integrated real-time intraoperative imaging, especially beneficial for procedures such as PN where quick and accurate suturing are essential for a successful outcome. Veeratterapillay et al. [1] present a series of 250 patients from centres in the UK, in which each performs <50 RAPN procedures/year, yet the authors present favourable outcomes overall, with a PSM rate of 7.3%, major complications in 6% and trifecta in 68.4%. An impressive learning curve is seen with improving outcomes over the series, such that in the final 50 cases a trifecta (WIT <25 min, negative surgical margin and absence of complications) was achieved in 82% of cases, with a PSM rate of 2% despite increasing complex nephrometry scores, which compares favourably with larger series from internationally renowned centres [6].
So then, with the results of the present study [1], can we say that Novick’s requirements have been met, and that minimally invasive NSS is now a ‘completely integrated option’? Certainly, with the widespread adoption of robot-assisted surgery, high-quality outcomes are within the grasp of centres other than elite academic institutions. As techniques develop and experience grows robot-assisted surgery can be increasingly offered, even for resection of more complex tumours.
To ensure that minimally invasive NSS is delivered to the highest standards, it will be necessary for providers to ensure both quality assurance and quality control in their processes. The learning curve needs to be minimised with structured teaching and mentoring, and the use of adjuncts such as intraoperative ultrasonography or fluorescence should be a routine part of care.
Centres offering this technique should be mindful of the well documented volume–outcome relationship that appears to be ubiquitous among complex surgical procedures. If centres are performing less than an optimum number of cases, they may consider affiliating themselves with other such centres in networks and forming a joint clinical governance programme, as has been described for robot-assisted radical prostatectomy and which has shown demonstrable improvements in outcomes.
Finally, auditing and reporting of outcomes remains the cornerstone of quality assurance as shown by the introduction of the BAUS complex surgery audit, which is intended to drive standards of care forward. Publications such as that of Veeratterapillay et al. [1] greatly assist in documenting the progress of new techniques and emerging technologies. Increasingly, patients expect transparency from healthcare providers, and with the necessary support processes in place, such initiatives, and the data that they produce will help to further improve the delivery of complex surgery to patients from all areas of our practice.
Article of the Week: PADUA and R.E.N.A.L. nephrometry scores correlate with perioperative outcomes of RAPN: analysis of the Vattikuti GQI-RUS database
Every week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
PADUA and R.E.N.A.L. nephrometry scores correlate with perioperative outcomes of robot-assisted partial nephrectomy: analysis of the Vattikuti Global Quality Initiative in Robotic Urologic Surgery (GQI-RUS) database
Abstract
Objectives
To evaluate and compare the correlations between Preoperative Aspects and Dimensions Used for an Anatomical (PADUA) and R.E.N.A.L. [Radius (tumour size as maximal diameter), Exophytic/endophytic properties of the tumour, Nearness of tumour deepest portion to the collecting system or sinus, Anterior (a)/posterior (p) descriptor and the Location relative to the polar line] nephrometry scores and perioperative outcomes and postoperative complications in a multicentre, international series of patients undergoing robot-assisted partial nephrectomy (RAPN) for masses suspicious for renal cell carcinoma (RCC).
Patients and Methods
We retrospectively evaluated the clinical records of patients who underwent RAPN between 2010 and 2013 for clinical N0M0 renal tumours in four international centres that completed all the data required for the Vattikuti Global Quality Initiative in Robotic Urologic Surgery (GQI-RUS) database. All patients underwent preoperative computed tomography or magnetic resonance imaging to define the clinical stage and anatomical characteristics of the tumours. PADUA and R.E.N.A.L. scores were retrospectively assessed in each centre. Univariate and multivariate analyses were used to evaluate the correlations between age, gender, Charlson comorbidity index, clinical tumour size, PADUA and R.E.N.A.L. complexity group categories and warm ischaemia time (WIT) of >20 min, urinary calyceal system closure, and grade of postoperative complications.
Results
Overall, 277 patients were evaluated. The median (interquartile range) tumour size was 33.0 (22.0–43.0) mm. The median PADUA and R.E.N.A.L. scores were eight and seven, respectively; 112 (40.4%), 86 (31.0%) and 79 (28.5%) patients were classified in the low-, intermediate- or high-complexity group according to PADUA score, while 118 (42.5%), 139 (50.1%) and 20 (7.2%) were classified in the low-, intermediate- or high-complexity group according to R.E.N.A.L. score, respectively. Both nephrometry tools significantly correlated with perioperative outcomes at univariate and multivariate analyses.
Conclusion
A precise stratification of patients before PN is recommended to consider both the potential threats and benefits of nephron-sparing surgery. In our present analysis, both PADUA and R.E.N.A.L. were significantly associated with predicting prolonged WIT and high-grade postoperative complications after RAPN.
Editorial: Nephrometry scoring systems: valuable research tools, but can they be applied in daily clinical practice?
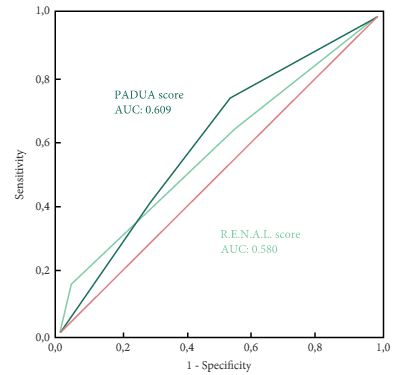
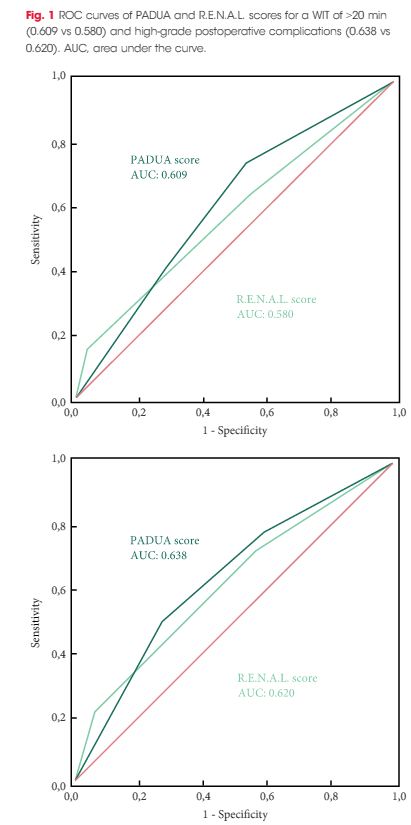
In this issue of BJUI Schiavina et al. [1] report on the RENAL and PADUA nephrometry scoring systems in predicting peri-operative outcomes, including warm ischaemia time and postoperative complications, in a multi-institutional cohort of patients undergoing robot-assisted partial nephrectomy. The authors showed that tumours classified as being of intermediate and high complexity on the PADUA score and high complexity on the RENAL score were associated with a nearly threefold higher risk of longer warm ischaemia times (>20 min). In addition, more complex tumours carried a higher risk of grade 3–4 postoperative complications (most commonly bleeding requiring angioembolization and urine leak requiring a ureteric stent). Notably, the two scoring systems were found to be similar predictors of these peri-operative outcomes on receiver-operating curve (ROC) analyses [1].
This represents the first large, multicentre study to evaluate the accuracy of these scoring systems in a cohort of patients who purely underwent robot-assisted surgery. A recent study by Borgmann et al. [2] found that, among the reported scoring systems, the RENAL nephrometry score correlated best with achieving negative surgical margins, shorter ischaemia times, and low postoperative complication rates; however, only 9% of patients underwent robot-assisted surgery. Another contemporary series showed concordance between the RENAL and PADUA scoring systems in predicting ischaemia times and complication rates, albeit in patients who only underwent open surgery [3].
Current guidelines recognize nephron-sparing approaches to small renal masses as the standard of care in well-selected patients, with the robot-assisted platform being predominantly adopted in clinical practice where available. Certainly, these nephrometry scores are valuable for urologists in counselling patients on the potential risk of complications specific to the surgical anatomy of the tumour. In addition, the RENAL and PADUA scores (and others) provide a quantitative, objective method for comparing data from different studies and different institutions.
As nephrometry scoring systems continue to be critically evaluated in the robotic surgery era, the question that naturally arises is: which system is best? With regard to this question, the data in the present study do not necessarily favour one or the other for the prediction of clinically relevant peri-operative outcomes. One must recognize, however, that several other anatomy-based scoring systems exist and were not examined in this manuscript [4-6]. While these are very valuable research and patient counselling tools, one must caution against using these nephrometry tools to make clinical decisions; for example, attempting to predict benign vs malignant histology (without a biopsy), attempting to predict high vs low grade tumours, or deciding on whether to perform a radical vs partial nephrectomy, or an open vs minimally invasive approach. After all, one must keep in mind that the area under the curve for these tools is in the range of 0.58–0.63 (0.50 being equivalent to toss of a coin).
It would have been interesting to include clinical size only in the present multivariate analysis (as was done for RENAL and PADUA scoring) and ROC analysis to compare this simple variable with the studied nephrometry scores. Future research should examine additional confounders that could potentially affect postoperative complication rates, such as BMI, adherent perinephric fat, experience of the surgeon actually performing the partial nephrectomy, technique of resection used (e.g. enucleation or resection) among others. This may help to distinguish a single system as the optimum model for use in research and in patient counselling regarding potential postoperative complications.
RE: Outcomes of high complex renal tumor (PADUA ≥ 10) following robot-assisted partial nephrectomy with a median 46 months follow-up: A tertiary center experience
Letter to the Editor
Outcomes of high complex renal tumor (PADUA ≥ 10) following robot-assisted partial nephrectomy with a median 46 months follow-up: A tertiary center experience
Sir,
We read this article by Raheem et al with great interest and appreciate the efforts of the authors to publish the largest single centre data on outcomes of high complexity tumors with PADUA score>10 [1]. We wish to highlight a few points. In table 1 the T stage classification has been applied to all the tumors irrespective of their benign or malignant nature. The AJCC TNM classification is particularly meant to stage renal cell carcinomas which are histopathologically proven [2].
It is clear from table 1 itself that there are 36 patients of Angiomyolipomas (AML) in the series which have erroneously been assigned a T stage and that it is likely that there may be other benign pathology in the patient cohort which might need to be de-staged once the pathology is available. Although this redistribution of staging might not affect the results, it is necessary so that the readers are not given a false impression of it being the same as well as to prevent the reproduction of such errors in future studies. A better way to categorize this can be seen in the study by Ficcara et al with benign tumors being categorized separately [3].
Another point of contention is that Fuhrman grading in table 1 has been applied to all the malignant tumors. For example in low PADUA score groups the total number of patients having Fuhrman grading is 52 while the number of patients with clear cell carcinoma is 42 with others being 5 in numbers. AML, papillary and chromphobe make up 25/72 tumors. It is known from the available literature that Fuhrman grading is only applied to clear cell carcinoma and its variants while Chromophobe carcinoma or papillary carcinoma is not graded by Fuhrman’s grading [4,5]. These points might not affect the basic theme of the study but are worth consideration.
Aditya Prakash Sharma. MBBS, MS
Senior Resident (M.Ch.), Department of Urology , PGIMER, Chandigarh
Girdhar S Bora, MS,M.Ch
Assistant professor, Department of Urology , PGIMER, Chandigarh
Ravimohan S Mavuduru, MS, M.Ch.
Associate Professor, Department of Urology , PGIMER, Chandigarh
Arup Kumar Mandal, MS, M.Ch.
Professor, Department of Urology, PGIMER, Chandigarh
References
- Abdel Raheem A, Alatawi A, Kim DK, Sheikh A, Alabdulaali I, Han WK, Choi YD, Rha KH. Outcomes of high-complexity renal tumours with a Preoperative Aspects and Dimensions Used for an Anatomical (PADUA) score of ≥10 after robot-assisted partial nephrectomy with a median 46.5-month follow-up: a tertiary centre experience. BJU Int. 2016 Apr 22. doi: 10.1111/bju.13501. [Epub ahead of print]
- Guinan, P., Sobin, LH, Algaba, F., Badellino, F., Kameyama, S., MacLennan, G. and Novick, A.(1997), TNM staging of renal cell carcinoma. Cancer, 80: 992–993
- Ficarra V, Novara G, Secco S, Macchi V, Porzionato A, De Caro R, Artibani W. Preoperative aspects and dimensions used for an anatomical (PADUA) classification of renal tumours in patients who are candidates for nephron-sparing surgery. Eur Urol. 2009 Nov;56(5):786-93.
- Delahunt B. Advances and controversies in grading and staging of renal cell carcinoma. Mod Pathol. 2009 Jun;22 Suppl 2:S24-36
- Sika-Paotonu D1, Bethwaite PB, McCredie MR, William Jordan T, Delahunt B. Nucleolar grade but not Fuhrman grade is applicable to papillary renal cell carcinoma. Am J Surg Pathol. 2006 Sep;30(9):1091-6.
Reply by the authors
We would like to thank the authors for their interest in reading our manuscript discussing the outcomes of high-complex PADUA renal tumors following robot-assisted partial nephrectomy (RAPN) [1]. We admit the discordance after pathological confirmation of benign and malignant nature of the masses and its need to be de-staged and revisited.
The main primary outcome of our study was to assess trifecta achievement and its predictors; meanwhile, the secondary end point was oncological safety and functional outcomes evaluation in patients with high-complex PADUA renal tumors. Notably, trifecta “i.e. WIT of <25 min, negative surgical margins and no and absence of perioperative complications” [2] does not incorporate long-term outcomes assessment after partial nephrectomy surgery; however, it provides us with an important data about the intraoperative surgical quality and efficiency, and early postoperative morbidity. PADUA classification is based on ‘preoperative radiological scoring of the renal masses’ and is applied for both benign and malignant masses [3].
In the current study, the majority of high-complex PADUA masses were malignant, and this group showed significant increase in conversion to radical nephrectomy, more perioperative morbidities, median WIT of 26 min; and subsequently, had a lower rate of trifecta achievement [1]. Previously, we reported excellent perioperative outcomes of angiomyolipoma (AML) after RAPN [4]. There were no intraoperative complications or blood transfusion. Moreover, WIT was short (median 19.5 min) [4]. Additionally, the long-term outcomes, AML has been proven to have good postoperative renal function preservation and no local recurrence after RAPN [4]. Putting in consideration the abovementioned results, it is apparent that inclusion of benign masses will not affect the outcomes.
Ali Abdel Raheem†*, Atalla Alatawi*, Dae Keun Kim¥, Abulhasan Sheikh*, Ibrahim Alabdulaali*, Woong Kyu Han*, Young Deuk Choi*, and Koon Ho Rha*
*Department of Urology and Urological Science Institute, Yonsei University College of Medicine, Seoul, South Korea
†Department of Urology, Tanta University Medical School, Egypt
¥Department of Urology, CHA Seoul Station Medical Center, CHA University Medical School, Seoul, Republic of Korea
References
- Abdel Raheem A, Alatawi A, Kim DK, et al. Outcomes of high-complexity renal tumours with a Preoperative Aspects and Dimensions Used for an Anatomical (PADUA) score of ≥10 after robot-assisted partial nephrectomy with a median 46.5-month follow-up: a tertiary centre experience. BJU Int. 2016 Apr 22. doi: 10.1111/bju.13501. [Epub ahead of print]
- Ficarra V, Novara G, Secco S et al. Perioperative aspects and dimensions used for an anatomical (PADUA) classification of renal tumours in patients who are candidates for nephron-sparing surgery. Eur Urol 2009;56: 786–93
- Khalifeh A, Autorino R, Hillyer SP et al. Comparative outcomes and assessment of trifecta in 500 robotic and laparoscopic partial nephrectomy cases: a single surgeon experience. J Urol 2013; 189: 1236–42
- Abdel Raheem A and Rha KH. RE: Robotic Partial Nephrectomy in the Treatment of Renal Angiomyolipomas. J Endourol.2016 Apr 15. [Epub ahead of print]
Article of the week: Repeat RAPN: Feasibility and early outcomes
Every week the Editor-in-Chief selects the Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Repeat robot-assisted partial nephrectomy (RAPN): feasibility and early outcomes
Riccardo Autorino, Ali Khalifeh, Humberto Laydner, Dinesh Samarasekera, Emad Rizkala, Remi Eyraud, Georges-Pascal Haber, Robert J. Stein and Jihad H. Kaouk
Center for Laparoscopic and Robotic Surgery, Glickman Urological and Kidney Institute, Cleveland Clinic, Cleveland, OH, USA
OBJECTIVE
• To demonstrate the feasibility, and to report our single-centre perioperative outcomes of repeat robot-assisted partial nephrectomy (RAPN).
PATIENTS AND METHODS
• From June 2006 to June 2012, 490 patients underwent RAPN for a renal mass at our centre. Of these patients, nine who had undergone previous ipsilateral nephron-sparing surgery (NSS) were included in the analysis.
• Patient charts were reviewed to obtain demographic data, preoperative surgical history, operative details, and postoperative outcomes and follow-up data.
RESULTS
• In all, 12 tumours were removed in nine patients (median age 69 years; six female). A third of the operations were performed on patients with a solitary kidney. The median (range) R.E.N.A.L. nephrometry score for the resected masses was 7 (4–8).
• The warm ischaemia time was 17.5 min and in three of the nine patients an unclamped procedure was performed. No intraoperative complications were registered, whereas only two minor complications occurred postoperatively. There were no renal unit losses. All surgical margins were negative.
• There was no significant difference between mean preoperative and latest postoperative mean estimated glomerular filtration rates (70.5 vs 63.5 mL/min/1.73m2, P > 0.05).
• At a mean (sd) follow-up of 8.3 (13) months, eight of the nine patients with a pathology diagnosis of malignant neoplasm were alive and free from disease at the latest follow-up.
CONCLUSION
• Although technically more demanding, repeat RAPN can be safely and effectively performed in patients presenting with local recurrence after primary NSS for kidney cancer.
Read Previous Articles of the Week