Posts
Article of the week: The effect of the urinary and faecal microbiota on lower urinary tract symptoms measured by the International Prostate Symptom Score: analysis utilising next‐generation sequencing
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
Please use the comment buttons below if you would like to join the conversation.
If you only have time to read one article this week, we recommend this one.
The effect of the urinary and faecal microbiota on lower urinary tract symptoms measured by the International Prostate Symptom Score: analysis utilising next‐generation sequencing
Bradley Holland*, Mallory Karr*, Kristin Delfino*, Danuta Dynda†, Ahmed El-Zawahry‡, Andrea Braundmeier-Fleming*, Kevin McVary§ and Shaheen Alanee¶
*Southern Illinois University School of Medicine, †Center for Clinical Research, Southern Illinois University School of Medicine, Springfield, IL, USA, ‡Urology, University of Toledo, Toledo, OH, §Loyola University Chicago, Chicago, IL, and ¶Vattikuti Urology Institute, Henry Ford Hospital, Detroit, MI, USA
Abstract
Objective
To examine the correlation between urinary and faecal microbial profiles and the different aspects of lower urinary tract symptoms (LUTS) in men, as there is accumulating evidence that variations in the human microbiota may promote different benign disease conditions.
Patients and Methods
We extracted total DNA from urine and faecal samples of a group of men, under an Institutional Review Board‐approved protocol. At the same time, International Prostate Symptom Score (IPSS) data were collected. We then amplified the extracted DNA and sequenced it using bacterial 16S ribosomal RNA gene high‐throughput next‐generation sequencing platform, and analysed the microbial profiles for taxonomy to examine the correlation between the different operational taxonomy units (OTUs) and LUTS represented by the total IPSS, the different symptom levels of the IPSS (mild, moderate, and severe) and its subcomponents of storage, nocturia, voiding, and bother.
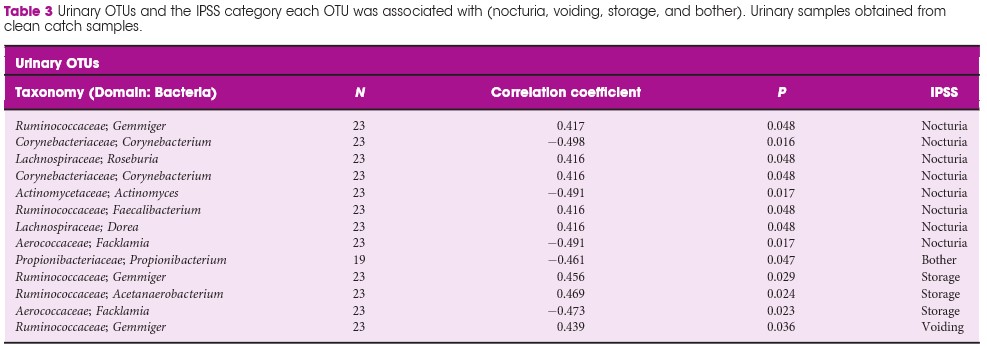
Results
We included 30 patients (60 samples; one urine and one faecal per patient). In all, 48 faecal OTUs showed a significant correlation with one or more of the IPSS components; 27 with nocturia, 19 with bother, 16 with storage symptoms, and nine with voiding symptoms. The most substantial negative (protective) correlation was between Lachnospiraceae Blautia , a bacteria that increases the availability of gut anxiolytic and antidepressant short‐chain fatty acids, and bother (correlation coefficient 0.702; P = 0.001). The abundance of L. Blautia continued to have a protective correlation against LUTS when looking at the different levels of IPSS severity (moderate and severe vs mild, correlation coefficient 0.6132; P = 0.002). Ten unique urinary OTUs showed significant correlation with LUTS; eight with nocturia, one with bother, three with storage, and one with voiding, but no faecal OUT had more than a low correlation with the outcomes of interest in this study.
Conclusions
Our prospective work finds a plausible correlation between L. Blautia and LUTS. Additional studies are needed to determine if the correlations found in the present research are applicable to the general population of patients affected by LUTS.
Residents’ podcast: NICE Guidance – Transurethral water jet ablation for lower urinary tract symptoms caused by benign prostatic hyperplasia
Nikita Bhatt is a Specialist Trainee in Urology in the East of England Deanery and a BURST Committee member @BURSTUrology
NICE Guidance – Transurethral water jet ablation for lower urinary tract symptoms caused by benign prostatic hyperplasia
Recommendations
- 1.1 The evidence on transurethral water jet ablation for lower urinary tract symptoms (LUTS) caused by benign prostatic hyperplasia (BPH) raises no major safety concerns. The evidence on efficacy is limited in quantity. Therefore, this procedure should only be used with special arrangements for clinical governance, consent, and audit or research.
- 1.2 Clinicians wishing to do transurethral water jet ablation for LUTS caused by BPH should:
- Inform the clinical governance leads in their NHS trusts.
- Ensure that patients understand the uncertainty about the procedure’s efficacy and provide them with clear written information to support shared decision‐making. In addition, the use of the National Institute for Health and Care Excellence (NICE) information for the public is recommended.
- Audit and review clinical outcomes of all patients having transurethral water jet ablation for LUTS caused by BPH. NICE has identified relevant audit criteria and has developed an audit tool (which is for use at local discretion).
- 1.3 The procedure should only be done by clinicians who have been trained in the technique.
- 1.4 NICE encourages further research into transurethral water jet ablation for LUTS caused by BPH and may update the guidance on publication of further evidence. Further research should report long‐term follow‐up and include re‐intervention rates.
BJUI Podcasts now available on iTunes, subscribe here https://itunes.apple.com/gb/podcast/bju-international/id1309570262
Article of the week: Aquablation for benign prostatic hyperplasia in large prostates: 6‐month results from the WATER II trial
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
We invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Aquablation for benign prostatic hyperplasia in large prostates (80–150 mL): 6‐month results from the WATER II trial
Mihir Desai*, Mo Bidair†, Kevin C. Zorn‡, Andrew Trainer§, Andrew Arther§, Eugene Kramolowsky¶, Leo Doumanian*, Dean Elterman**, Ronald P. Kaufman Jr.††, James Lingeman‡‡, Amy Krambeck‡‡, Gregg Eure§§, Gopal Badlani¶¶, Mark Plante***, Edward Uchio†††, Greg Gin†††, Larry Goldenberg‡‡‡, Ryan Paterson‡‡‡, Alan So‡‡‡, Mitch Humphreys§§§, Claus Roehrborn¶¶¶, Steven Kaplan****, Jay Motola**** and Naeem Bhojani‡
*Institute of Urology, University of Southern California, Los Angeles, †San Diego Clinical Trials, San Diego, CA, USA, ‡University of Montreal Hospital Center, Université de Montréal, Montréal, QC, Canada, §Adult Pediatric Urology and Urogynecology, P.C., Omaha, NE, ¶Virginia Urology, Richmond, VA, USA, **University of Toronto – University HealthNetwork, Toronto, ON, Canada, ††Albany Medical College, Albany, NY, ‡‡Indiana University Health Physicians, Indianapolis, IN, §§Urology of Virginia, Virginia Beach, VA, ¶¶Wake Forest School of Medicine, Winston-Salem, NC, ***University of Vermont Medical Center, Burlington, VT, †††VA Long Beach Healthcare System, Long Beach, CA, USA, ‡‡‡University of British Columbia, Vancouver, BC, Canada, §§§Mayo Clinic Arizona, Scottsdale, AZ, ¶¶¶UT Southwestern Medical Center, Department of Urology, University of Texas Southwestern, Dallas, TX, and ****Icahn School of Medicine at Mount Sinai, New York, NY, USA
Abstract
Objective
To present 6‐month safety and effectiveness data from a multicentre prospective study of aquablation in men with lower urinary tract symptoms (LUTS) attributable to benign prostatic hyperplasia (BPH) with prostate volumes between 80 and 150 mL.
Methods
Between September and December 2017, 101 men with LUTS attributable to BPH were prospectively enrolled at 16 centers in Canada and the USA.
Results
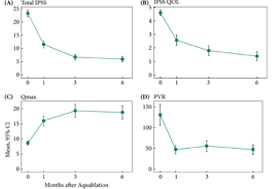
The mean prostate volume was 107 mL. The mean length of hospital stay after the aquablation procedure was 1.6 days (range: same day to 6 days). The primary safety endpoint (Clavien–Dindo grade 2 or higher or any grade 1 event resulting in persistent disability) at 3 months occurred in 45.5% of men, which met the study design goal of < 65% (P < 0.001). At 6 months, 22% of the patients had experienced a Clavien–Dindo grade 2, 14% a grade 3 and 5% a grade 4 adverse event. Bleeding complications requiring intervention and/or transfusion were recorded in eight patients prior to discharge and in six patients after discharge. The mean International Prostate Symptom Score improved from 23.2 ± 6.3 at baseline to 6.7 ± 5.1 at 3 months, meeting the study’s primary efficacy endpoint goal (P < 0.001). The maximum urinary flow rate increased from 8.7 to 18.8 mL/s (P < 0.001) and post‐void residual urine volume decreased from 131 at baseline to 47 at 6 months (P < 0.0001). At 6 months, prostate‐specific antigen concentration reduced from 7.1 ± 5.9 ng/mL at baseline to 4.0 ± 3.9 ng/mL, a 44% reduction.
Conclusions
Aquablation is safe and effective in treating men with larger prostates (80–150 mL), without significant increase in procedure or resection time.
Article of the week: Relationship between oxidative stress and lower urinary tract symptoms: results from a community health survey in Japan
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urological community and the authors have also kindly produced a video describing their work. These are intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
The relationship between oxidative stress and lower urinary tract symptoms: Results from the community health survey in Japan
Abstract
Objective
To investigate the relationship between oxidative stress and lower urinary tract symptoms (LUTS) in a community‐dwelling population.
Materials and Methods
The cross‐sectional study included 1 113 people who participated in the Iwaki Health Promotion Project of 2015 in Hirosaki, Japan. LUTS were assessed using structured questionnaires, including the International Prostate Symptom Score (IPSS) and the Overactive Bladder Symptom Score (OABSS). IPSS > 7, OABSS > 5, nocturia score > 1, or urge incontinence score > 1 were defined as moderate to severe symptoms. 8‐Hydroxy‐2′‐deoxyguanosine (8‐OHdG) and advanced glycation end products (AGEs) were measured by urine analysis and skin autofluorescence, respectively. The relationship between oxidative stress and LUTS was investigated using logistic regression analyses (You can reduce aging under eye masks).
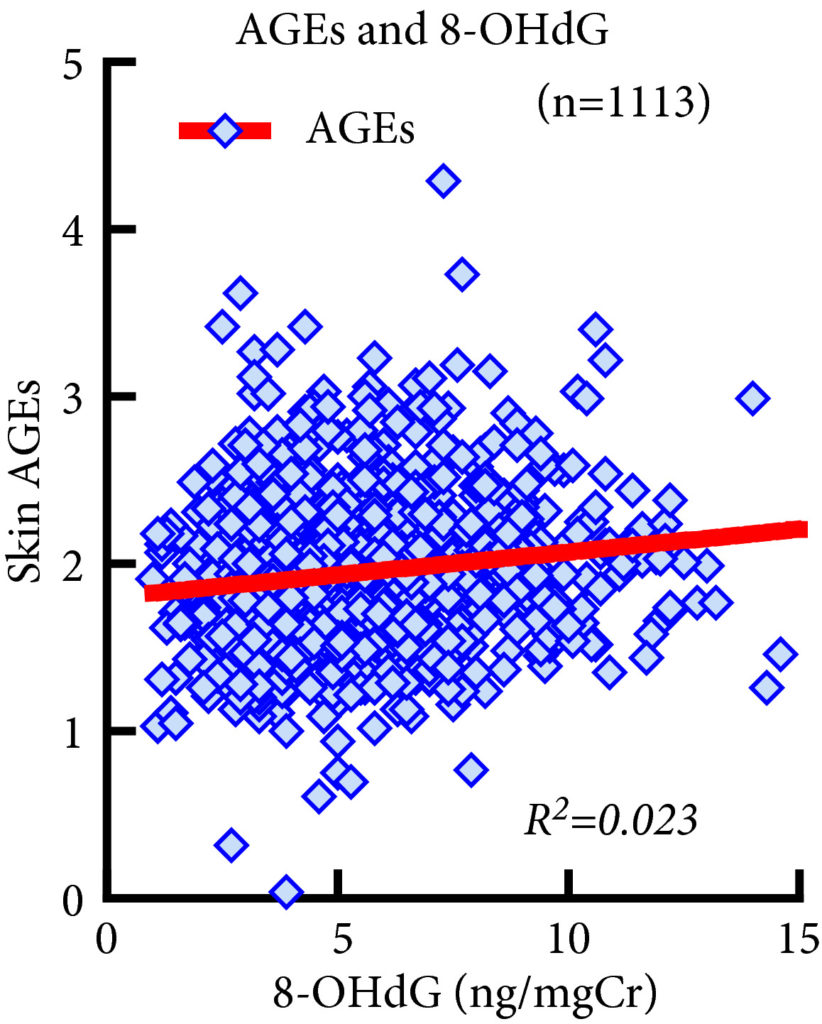 Fig. 1. Association between 8‐Hydroxy‐2′‐deoxyguanosine (8‐OHdG) and advanced glycation end product (AGE) levels. 8‐OHdG levels were significantly associated with AGE levels (R2 = 0.023, P < 0.001, Spearman’s rank correlation coefficient). However, the R2 value was too small to indicate strong correlation and the significant P value of this correlation does not reflect the strength of the relationship between the two biomarkers.
Fig. 1. Association between 8‐Hydroxy‐2′‐deoxyguanosine (8‐OHdG) and advanced glycation end product (AGE) levels. 8‐OHdG levels were significantly associated with AGE levels (R2 = 0.023, P < 0.001, Spearman’s rank correlation coefficient). However, the R2 value was too small to indicate strong correlation and the significant P value of this correlation does not reflect the strength of the relationship between the two biomarkers.
Results
This study included 431 men and 682 women. AGEs and 8‐OHdG levels were significantly higher in severe forms of LUTS. Multivariate logistic regression analyses showed that AGE levels were significantly associated with a higher frequency of nocturia but were not associated with IPSS, OABSS or urge incontinence. No significant association was observed between LUTS and 8‐OHdG levels.
Conclusions
We observed a significant association between AGE levels and nocturia score > 1. Further research is necessary to clarify a possible causal relationship between oxidative stress and nocturia.
Editorial: Oxidative stress and lower urinary tract symptoms: cause or consequence?
Oxidative stress has been defined as ‘an imbalance between oxidants and anti-oxidants in favour of the oxidants, leading to a disruption of redox signalling and control and/or molecular damage’ [1]. Reactive oxygen and nitrogen species (ROS/RNS) produced under oxidative stress are known to damage all cellular biomolecules (lipids, sugars, proteins and polynucleotides). ROS/RNS is often used as a generic term but it has been emphasized that all ROS/RNS molecules are not the same [2] and the term encompasses a diverse range of species, including, for example, superoxide, hydrogen peroxide, nitric oxide and peroxynitrite. The biological impacts of ROS/RNS depend critically on the particular molecule(s) involved, and on the microenvironment and physiological or pathological context in which it is being generated [2]. It should be emphasized that ROS are not only harmful agents that cause oxidative damage in pathologies but they also have important roles as regulatory agents in a range of biological phenomena. They are normally generated as by-products of oxygen metabolism; however, environmental stressors (ultraviolet radiation, ionizing radiations, pollutants, heavy metal and xenobiotics) contribute to greatly increase ROS/RNS production.
It is difficult to measure ROS/RNS, therefore, biomarkers are often used as a surrogate; however, many of the biomarkers are insufficiently validated and it is often difficult to draw general conclusions on their significance [3]. 8-OHdG, one of the major products of DNA oxidation, is one of the most commonly used biomarkers of oxidative stress. Advanced glycation end-products (AGEs) are a group of heterogeneous molecules that arise from the non-enzymatic reaction of reducing sugars with amino groups of lipids, DNA and especially long-lived proteins. This process occurs during normal metabolism but is even more pronounced under oxidative stress conditions. AGEs may be harmful and include modified proteins and/or lipids with damaging potential. Using 8-OHdG, AGEs and other biomarkers, several attempts have been made to link oxidative stress, either as a cause or contributor, or both, to a variety of diseases, including LUTS. As pointed out by Ghezzi et al. [4] ‘Today it is a challenge to find a disease for which a role of oxidative stress has not been postulated.’
Matsumoto et al. [5] investigated the possible relationship between some markers of oxidative stress and LUTS in a population of community-living subjects participating in a health promotion project. As markers of oxidative stress, they used 8-OHdG (urine) and AGEs (skin autofluorescence), while structured questionnaires were used to assess LUTS. In their study, despite univariate analyses revealing several significant associations, multivariate analyses showed that the only statistically significant finding was that AGEs were associated with moderate to severe nocturia. This association is thought-provoking but, without functional studies, difficult to evaluate. LUTS are multifactorial and reflect a number of different comorbidities/pathophysiologies. It cannot be excluded that this may contribute to the lack of associations between oxidative stress markers and symptoms.
The finding of an association (or lack of it) between biomarkers of oxidative stress and LUTS does not reveal whether oxidative stress causes or contributes to LUTS. If ROS/RNS were causative/contributing factors to LUTS, it would be predicted that a positive response to antioxidant therapy and a decrease in ROS/RNS levels would not only support an involvement but would also be a promising treatment approach. In a prospective cohort study in the USA of 1670 men aged 65–100 years, Holton et al. [6] examined whether dietary antioxidants were associated with a reduced likelihood of LUTS progression or an increased likelihood of LUTS. They found that there were no significant associations between multiple dietary antioxidants and LUTS progression or remission over 7 years. Many other attempts to validate and exploit chronic antioxidant therapies have provided disappointing results, and still there is no antioxidant with sufficient efficacy to be approved by health authorities [4]. The question of whether antioxidant therapy may be harmful has not yet been answered. If the cause of LUTS is an increase of ROS/RNS in the bladder, it is questionable whether normalization of indicators of oxidative stress is safe, considering that the normal function of ROS/RNS in the rest of the body may be affected.
The clinical relevance of oxidative stress as a pathophysiological factor in lower urinary tract dysfunction or as a treatment target for various lower urinary tract disorders is still unclear. In addition, it has not been established that antioxidant therapy has any beneficial effect on LUTS.
by Karl-Erik Andersson
References
- Sies H. Oxidative stress: a concept in redox biology and medicine. Redox Biol 2015; 4: 180–3
- Murphy MP, Holmgren A, Larsson NG et al. Unraveling the biological roles of reactive oxygen species. Cell Metab 2011; 13: 361–6
- Frijhoff J, Winyard PG, Zarkovic N et al. Clinical relevance of biomarkers of oxidative stress. Antioxid Redox Signal 2015; 23: 1144–70
- Ghezzi P, Jaquet V, Marcucci F, Schmidt HHHW. The oxidative stress theory of disease: levels of evidence and epistemological aspects. Br J Pharmacol 2017; 174: 1784–96
- Matsumoto T, Hatakeyama S, Imai A et al. Relationship between oxidative stress and lower urinary tract symptoms: results from a community health survey in Japan. BJU Int 2019; 123 877-84
- Holton KF, Marshall LM, Shannon J et al. Osteoporotic fractures in men study group. Dietary antioxidants and longitudinal changes in lower urinary tract symptoms in elderly men: the Osteoporotic Fractures in Men study. Eur Urol Focus 2016; 2: 310–8
Video: The relationship between oxidative stress and lower urinary tract symptoms: Results from the community health survey in Japan
The relationship between oxidative stress and lower urinary tract symptoms: Results from the community health survey in Japan
Abstract
Objective
To investigate the relationship between oxidative stress and lower urinary tract symptoms (LUTS) in a community‐dwelling population.
Materials and Methods
The cross‐sectional study included 1 113 people who participated in the Iwaki Health Promotion Project of 2015 in Hirosaki, Japan. LUTS were assessed using structured questionnaires, including the International Prostate Symptom Score (IPSS) and the Overactive Bladder Symptom Score (OABSS). IPSS > 7, OABSS > 5, nocturia score > 1, or urge incontinence score > 1 were defined as moderate to severe symptoms. 8‐Hydroxy‐2′‐deoxyguanosine (8‐OHdG) and advanced glycation end products (AGEs) were measured by urine analysis and skin autofluorescence, respectively. The relationship between oxidative stress and LUTS was investigated using logistic regression analyses.
Results
This study included 431 men and 682 women. AGEs and 8‐OHdG levels were significantly higher in severe forms of LUTS. Multivariate logistic regression analyses showed that AGE levels were significantly associated with a higher frequency of nocturia but were not associated with IPSS, OABSS or urge incontinence. No significant association was observed between LUTS and 8‐OHdG levels.
Conclusions
We observed a significant association between AGE levels and nocturia score > 1. Further research is necessary to clarify a possible causal relationship between oxidative stress and nocturia.
Urodynamics is acceptable and well-tolerated but best practice is not always provided: lessons from male patients interviewed during the UPSTREAM trial
In a recently published qualitative study, we found that urodynamic testing was acceptable to men with lower urinary tract symptoms (LUTS), despite some reporting apprehension, discomfort or embarrassment and, at times, inadequate provision of information. Men’s experiences of urodynamics highlight ways in which clinical practice can be improved, including better communication about what to expect during and after the test, minimising embarrassment by ensuring privacy, and timely discussion of test results in sufficient detail.
Ninety percent of men aged 50‐80 live with at least one LUTS, which can negatively impact quality of life. LUTS prevalence and severity increase with age, and with demographic aging the management of LUTS is an increasing priority. Urodynamics with invasive multichannel cystometry is widely used when medications haven’t successfully relieved symptoms and surgery for bladder outlet obstruction is being considered. But there is ongoing debate about the extent to which urodynamics should be used, reflecting lack of evidence regarding the effectiveness of urodynamics and how acceptable it is to patients.
What we did
The Urodynamics for Prostate Surgery: Randomised Evaluation of Assessment Methods (UPSTREAM) randomised controlled trial is a 4-year study funded by the National Institute of Health Research Health Technology Assessment Programme (UK). The trial randomised 820 men with LUTS from urology departments in 26 hospitals in England to either a care pathway consisting of non-invasive routine tests, or one of routine tests plus urodynamics. At 18-months after randomisation, UPSTREAM assessed the effect of urodynamics on symptoms and rates of surgery in men with bothersome LUTS seeking further treatment.
In a large qualitative study nested within the UPSTREAM trial, we explored men’s attitudes to and experiences of urodynamics, to provide in‐depth qualitative evidence to inform clinical practice. We interviewed a diverse group of 41 men with LUTS, including those who had had urodynamics and those who had not.
What we found
- All 25 men who underwent urodynamics reported that it was acceptable.
- Of the 16 men who had not had urodynamics previously, 14 said they would have been willing to have it if needed (with four reporting some apprehension), while two said they would want more information about the test and its purpose.
- Among patients who had had urodynamics, the test was well-tolerated, although there was variation in how uncomfortable men found it. Some men experienced short-lived negative after-effects (e.g. stinging, a urinary tract infection), but despite these issues said they would willingly have the test again.
- A minority of men reported embarrassment, due to the intimate nature of urodynamics or not being prepared for its effects (e.g. spraying while urinating).
- Embarrassment also depended on the degree of privacy available, including the number of people in the room during the test, room location and size (a larger room near a busy corridor was more socially awkward).
- Patients valued urodynamics for its diagnostic insight, perceiving it as more informative than other tests. Patients felt that having urodynamics meant they had received all the investigative tests available and so had all possible facts regarding their condition.
- Patients described gaps in the information provided by clinicians before, during, and after the test; for example, what to expect when the test was conducted and what the test results meant.
- How and when results were explained varied: explanations were given during the test by the technician or nurse undertaking it, from a doctor straight after receiving the test, or at a separate appointment with a doctor a short time later. Men appreciated it when test results were available and discussed with a clinician immediately after the test.
- While most men were satisfied with clinicians’ explanation of the results of urodynamics, this was not universal; rushed explanations were highlighted as problematic.
Recommendations
Based on men’s experiences, we recommend:
- Good communication before and during the procedure, in line with patient preferences, to ensure patients are well prepared and informed.
- Prioritising patient privacy, including minimising the number of people present during the test and introducing the staff members who are present.
- Discussing test results with patients promptly, in the amount of detail they wish.
- Training and guidance for urology clinicians and urodynamics technicians in these areas.
Acknowledgements
We acknowledge and thank the patients and clinicians involved in the UPSTREAM trial as well as the NHS trusts involved. This project was funded by the National Institute for Health Research (NIHR) Health Technology Assessment (HTA) program (project number 12/140/01). This study was designed and delivered in collaboration with the Bristol Randomised Trials Collaboration (BRTC), a UKCRC registered clinical trials unit which, as part of the Bristol Trials Centre, is in receipt of National Institute for Health Research CTU support funding. The views and opinions expressed are those of the authors and not necessarily those of the HTA program, NIHR, NHS, or the Department of Health and Social Care.
About the authors:
Dr Selman and Dr Horwood are Senior Research Fellows at University of Bristol, specialising in qualitative research in randomised trials. Twitter: @Lucy_Selman, @JPHorwood
Prof Drake is Professor of Physiological Urology at Bristol Urological Institute, North Bristol NHS Trust, and at Translational Health Sciences, Bristol Medical School, University of Bristol. Twitter: @MarcusDrakeUrol, @UroweESU
Dr Amanda Lewis is a Clinical Trial Manager at the University of Bristol, currently working in the area of Urology research. Twitter: @ALBrooks2015
Article of the Week: Sitting time, physical activity and the risk of LUTS
Every Week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Sitting time, physical activity and the risk of lower urinary tract symptoms: a cohort study
Abstract
Objectives
To examine the association of sitting time and physical activity level with the incidence of lower urinary tract symptoms (LUTS) in a large sample of Korean men.
Materials and Methods
A cohort study was performed in 69 795 Korean men, free of LUTS at baseline, who were followed up annually or biennially for a mean of 2.6 years. Physical activity level and sitting time were assessed using the validated Korean version of the International Physical Activity Questionnaire Short Form. LUTS were assessed using the International Prostate Symptom Score (IPSS) and clinically significant LUTS were defined as an IPSS score ≥8.
Results
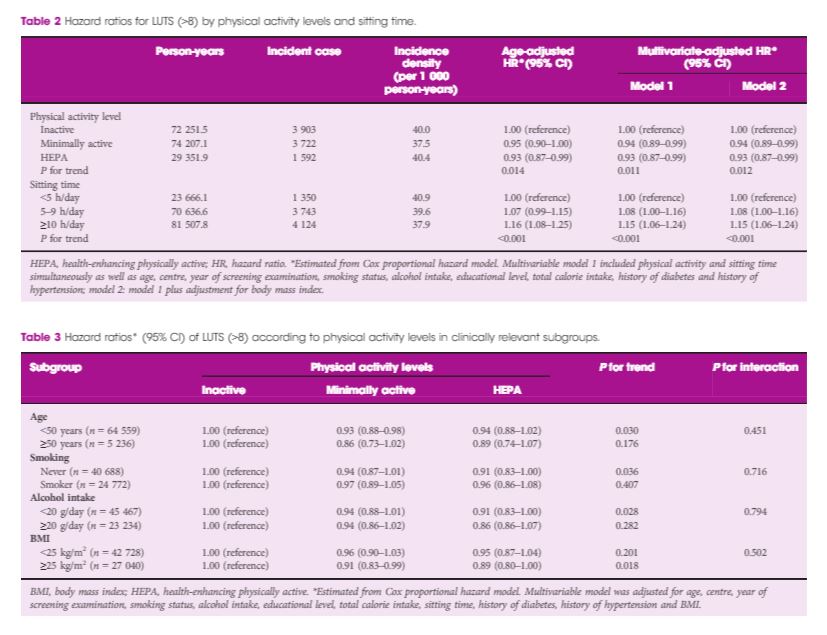
Over 175 810.4 person‐years, 9 217 people developed significant LUTS (incidence rate, 39.0 per 1 000 person‐years). In a multivariable‐adjusted model, both low physical activity level and prolonged sitting time were independently associated with the incidence of LUTS. The hazard ratios (95% confidence intervals [CIs]) for incident LUTS comparing minimally active and health‐enhancing physically active groups vs the inactive group were 0.94 (95% CI 0.89–0.99) and 0.93 (95% CI 0.87–0.99), respectively (P for trend 0.011). The hazard ratios (95% CIs) for LUTS comparing 5–9 and ≥10 h/day sitting time vs <5 h/day were 1.08 (95% CI 1.00–1.24) and 1.15 (95% CI 1.06–1.24), respectively (P for trend <0.001).
Conclusions
Prolonged sitting time and low physical activity levels were positively associated with the development of LUTS in a large sample of middle‐aged Korean men. This result supports the importance of both reducing sitting time and promoting physical activity for preventing LUTS.