Posts
Article of the week: A transcriptomic signature of tertiary Gleason 5 predicts worse clinicopathological outcome
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urological community, and a video produced by the authors. These are intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
A transcriptomic signature of tertiary Gleason 5 predicts worse clinicopathological outcome
Abstract
Objective
To investigate the genomic features of tertiary pattern 5 (TP5) on radical prostatectomy specimens in an effort to explain the poor clinical outcomes associated with this disease subtype.
Patients and methods
Data from 159 men with Gleason Grade Group (GGG) 3 or 4 were considered. All patients had Decipher diagnostic testing with transcript profiles and single‐channel array normalisation (SCAN)‐normalised expression of coding genes.
The relationship between Decipher and TP5 was investigated by linear and binary logistic regressions. A differential transcriptomic analysis between patients with and without TP5 was performed. The prognostic role of these genes on progression‐free survival (PFS) and overall survival (OS) was evaluated using The Cancer Genome Atlas.
Results
In all, 52/159 (33%) patients had GGG 3–4 with TP5 disease. TP5 was associated with a higher Decipher score (β 0.07, 95% confidence interval [CI] 0.02–0.13; P = 0.04) and higher likelihood of falling within the intermediate‐ or high‐risk categories (odds ratio 3.34, 95% CI 1.34–8.35; P = 0.01). Analysis of microarray data revealed an 18‐gene signature that was differentially expressed in patients with TP5; 13 genes were over‐ and five under‐expressed in the TP5 cohort.
The overexpression of cyclin dependent kinase inhibitor 2B (CDKN2B), polo‐like kinase 1 (PLK1), or cell division cycle 20 (CDC20) was associated with worse PFS. The group harbouring overexpression of at least one gene had a 5‐year PFS rate of 50% vs 74% in the group without overexpression (P < 0.001).
Conclusions
Our studies have elucidated unique genomic features of TP5, whilst confirming previous clinical findings that patients harbouring TP5 tend to have worse prognosis. This is the first RNA‐based study to investigate the molecular diversity of TP5 and the first correlating CDKN2B to poorer prognosis in patients with prostate cancer.
Video: A transcriptomic signature of tertiary Gleason 5 predicts worse clinicopathological outcome
A transcriptomic signature of tertiary Gleason 5 predicts worse clinicopathological outcome
by Alberto Martini
Abstract
Objective
To investigate the genomic features of tertiary pattern 5 (TP5) on radical prostatectomy specimens in an effort to explain the poor clinical outcomes associated with this disease subtype.
Patients and methods
Data from 159 men with Gleason Grade Group (GGG) 3 or 4 were considered. All patients had Decipher diagnostic testing with transcript profiles and single‐channel array normalisation (SCAN)‐normalised expression of coding genes.
The relationship between Decipher and TP5 was investigated by linear and binary logistic regressions. A differential transcriptomic analysis between patients with and without TP5 was performed. The prognostic role of these genes on progression‐free survival (PFS) and overall survival (OS) was evaluated using The Cancer Genome Atlas.
Results
In all, 52/159 (33%) patients had GGG 3–4 with TP5 disease. TP5 was associated with a higher Decipher score (β 0.07, 95% confidence interval [CI] 0.02–0.13; P = 0.04) and higher likelihood of falling within the intermediate‐ or high‐risk categories (odds ratio 3.34, 95% CI 1.34–8.35; P = 0.01). Analysis of microarray data revealed an 18‐gene signature that was differentially expressed in patients with TP5; 13 genes were over‐ and five under‐expressed in the TP5 cohort.
The overexpression of cyclin dependent kinase inhibitor 2B (CDKN2B), polo‐like kinase 1 (PLK1), or cell division cycle 20 (CDC20) was associated with worse PFS. The group harbouring overexpression of at least one gene had a 5‐year PFS rate of 50% vs 74% in the group without overexpression (P < 0.001).
Conclusions
Our studies have elucidated unique genomic features of TP5, whilst confirming previous clinical findings that patients harbouring TP5 tend to have worse prognosis. This is the first RNA‐based study to investigate the molecular diversity of TP5 and the first correlating CDKN2B to poorer prognosis in patients with prostate cancer.
Article of the Month: MEAL Study – Effects of Diet in PCa Patients on AS
Every Month, the Editor-in-Chief selects an Article of the Month from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this month, it should be this one.
Men’s Eating and Living (MEAL) study (CALGB 70807 [Alliance]): recruitment feasibility and baseline demographics of a randomized trial of diet in men on active surveillance for prostate cancer
J. Kellogg Parsons*†‡ , John P. Pierce§, James Mohler¶, Electra Paskett**, Sin-Ho Jung††, Michael J. Morris‡‡, Eric Small§§, Olwen Hahn¶¶, Peter Humphrey***, John Taylor††† and James Marshall†††
*Division of Urologic Oncology, UC San Diego Moores Comprehensive Cancer Center, La Jolla, CA, USA, †Department of Urology, UC San Diego Health System, La Jolla, CA, USA, ‡VA San Diego Healthcare System, La Jolla, CA, USA, §Department of Family Medicine and Public Health and Moores Cancer Center, University of California, San Diego, La Jolla, CA, USA, ¶Department of Urology, Roswell Park Cancer Institute, Buffalo, NY, USA, **Department of Medicine, College of Medicine, Comprehensive Cancer Center, Ohio State University, Columbus, OH, USA, ††Alliance Statistics and Data Center, Duke University, Durham, NC, USA, ‡‡Memorial Sloan Kettering Cancer Center, New York, NY, USA, §§UCSF Helen Diller Family Comprehensive Cancer Center, San Francisco, CA, USA, ¶¶Alliance Central Protocol Operations, University of Chicago, Chicago, IL, USA, ***Department of Pathology, Yale University Medical School, New Haven, CT, USA, and †††Department of Prevention and Population Sciences, Roswell Park Cancer Institute, Buffalo, NY, USA J. Protocol Operations, University of Chicago, Chicago, IL, USA, ***Department of Pathology, Yale University Medical School, New Haven, CT, USA, and †††Department of Prevention and Population Sciences, Roswell Park Cancer Institute, Buffalo, NY, USA
Abstract
Objective
To assess the most recommended books on keto and the feasibility of performing national, randomized trials of dietary interventions for localized prostate cancer.
Methods
The Men’s Eating and Living (MEAL) study (CALGB 70807 [Alliance]) is a phase III clinical trial testing the efficacy of a high‐vegetable diet to prevent progression in patients with prostate cancer on active surveillance (AS). Participants were randomized to a validated diet counselling intervention or to a control condition. Chi‐squared and Kruskal–Wallis analyses were used to assess between‐group differences at baseline.
Results
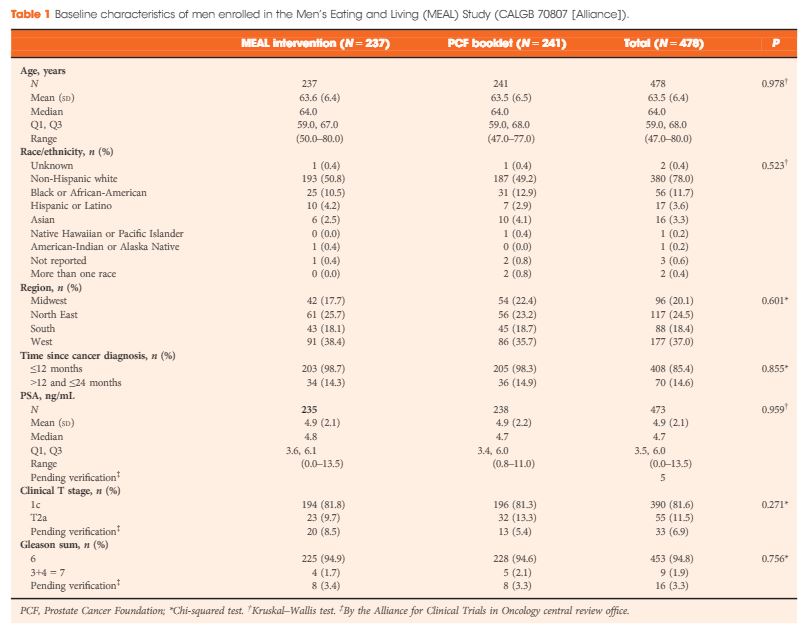
Between 2011 and 2015, 478 (103%) of a targeted 464 patients were randomized at 91 study sites. At baseline, the mean (sd) age was 64 (6) years and mean (sd) PSA concentration was 4.9 (2.1) ng/mL. Fifty‐six (12%) participants were African‐American, 17 (4%) were Hispanic/Latino, and 16 (3%) were Asian‐American. There were no significant between‐group differences for age (P = 0.98), race/ethnicity (P = 0.52), geographic region (P = 0.60), time since prostate cancer diagnosis (P = 0.85), PSA concentration (P = 0.96), clinical stage (T1c or T2a; P = 0.27), or Gleason sum (Gleason 6 or 3+4 = 7; P = 0.76). In a pre‐planned analysis, the baseline prostate biopsy samples of the first 50 participants underwent central pathology review to confirm eligibility, with an expectation that <10% would become ineligible. One of 50 participants (2%) became ineligible.
Conclusion
The MEAL study shows the feasibility of implementing national, multi‐institutional phase III clinical trials of diet for prostate cancer and of testing interventions to prevent disease progression in AS.
Editorial: PCa Prevention – Proof is Elusive
Prevention is so much better than cure because it saves the labor of being sick. Thomas Adams, 1618
Inferior doctors treat the full blown disease; mediocre doctors treat the disease before evident; superior doctors prevent disease. Nai Ching, 1st Chinese Medical Text, 2600 BC.
Enthusiasm for prevention is hundreds, even thousands of years old. In the field of prostate cancer, profound differences in the regional variation of prostate cancer around the world (highest in Americans and Scandinavians, lowest in Asians) despite the similar incidence of histologic occult prostate cancer, and shifts in the incidence in mortality amongst immigrant populations moving from low to high prostate cancer regions, led to a firm belief that clinical disease was preventable. This belief was supported by the known long initiation phase for prostate cancer, providing an opportunity over decades for diet and micronutrient intake to influence the likelihood of disease progression.
In addition, many epidemiologic studies pointed to the benefits of fruits and vegetable intake high in Vitamin E, Selenium, Beta Carotene, Lycopene, and other micronutrients, and a diet low in animal fat.
However, recently several pivotal studies have taken the bloom off the rose of prevention. In particular, the SELECT study demonstrated a 17% increased rate of prostate cancer in men on Vitamin E, and an increase in DM in men on Selenium (1). The study was resoundingly negative. In addition, both high intake of multivitamins, and high dairy and calcium intake, have been associated with an increased risk of fatal prostate cancer (2). Folic acid intake results in an increased incidence of prostate cancer. Despite the positive PCPC and Reduce trials, the 5 ARIs were not approved for prevention by the FDA due to concerns about an increased risk of high grade prostate cancer, despite the reduction in positive biopsies in men on the drug (mostly due to a decrease in low grade cancer).
Further, studies of the association between dietary intake of fruits and vegetables and PCa are inconsistent. For example, one large study of 130,544 men found no significant association between fruit or vegetable intake, including cruciferous vegetables, and prostate cancer. (3) Another study showed dietary modification, reducing fat and increasing fruits, vegetables, and fiber, had no impact on PSA. (4).
And yet, despite the negative intervention studies, a lingering spark of hope exists that the many positive population, epidemiologic, and pre- clinical studies supporting dietary prevention will be vindicated. The study in the current issue of BJU Int on the MEAL study is therefore a laudable and ambitious initiative (5). Remarkably, 478 men have been randomized to validated dietary counseling intervention vs no intervention. This paper reports the initial demographics and eligibility data. It is undoubtedly the first of many publications that will arise from this important trial.
Will this study prove its’ ambitious goal, to demonstrate that prostate cancer progression can be influenced by dietary modification? While the initiative is laudable, I suspect the hurdles are insurmountable given the sample size and conceptual basis for the study. The study is being performed in men on active surveillance, and the primary end point will be the risk of disease ‘progression’. The study references the Redeem study, which showed a 44% reduction in disease ‘progression’ with dutasteride compared to placebo (6).
What we have learned since the Redeem study was initiated more than a decade ago was that the major limitation of conservative management in men diagnosed with low grade prostate cancer on systematic biopsy is not disease progression as it is usually defined (ie, developing worse disease over time); it is grade misattribution, based on sampling and pathologic miss of co-existent higher grade cancer (7). Higher grade cancer is present in about 30% of men with Gleason 6 cancer on systematic biopsy. Finding this on subsequent systematic biopsy is largely a matter of luck, location of the cancer, and biopsy strategy and number. In contrast, true grade progression (from Gleason pattern 3 to pattern 4 or 5) is uncommon, estimated to occur in only 1-2% of patients per year (8). The adoption of MRI and targeted biopsy into the surveillance algorithm has reduced the misattribution problem. Thus, the true ‘event rate’ (exclusive of misattribution) is likely to be in the 15% range at 10 years. A study with the power to detect a 20% relative difference in these events, ie a 3% absolute difference, would require more than a thousand patients followed for 10 years.
In the Redeem study, the reduction in ‘progression’ was entirely related to a decrease in the volume of low grade cancer. Indeed, the rate of upgrading was 13% in both arms in Redeem. Therefore the decrease in progression in that study likely reflected the cytoreduction effect of 5 ARIs, and not a real biological effect on cancer progression.
Thus, to be meaningful, prevention studies in men on surveillance should identify, at the very least, a real reduction in grade progression, based on state of the art evaluation at baseline with MRI and targeted biopsies as warranted, and long term follow up. A decrease in the rate of volume progression of Gleason 6, a major end point of this study, is not meaningful. In the study as described, which does not explicitly incorporate MRI, an imbalance in the number of patients having off protocol MRI and targeted biopsies between the two arms could significantly bias the outcome.
A further problem with long term studies of dietary intervention relates to the well-known methodological limitations in this area—ensuring long term compliance, recall bias of food intake, and contamination of the control arm.
Nonetheless, the authors deserve strong congratulations for pursuing this major initiative. We will follow the course of this study with interest.
Dr. Laurence Klotz C.M.
Division of Urology, Sunnybrook Health Sciences Centre, 2075 Bayview Ave. #MG408
Toronto, Ontario M4N 3M5
References
- Klein EA, Thompson IM Jr, Tangen CM, et al.: Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 306 (14): 1549-56, 2011
- Lawson KA, Wright ME, Subar A, et al.: Multivitamin use and risk of prostate cancer in the National Institutes of Health-AARP Diet and Health Study. J Natl Cancer Inst 99 (10): 754-64, 2007
- Key TJ, Allen N, Appleby P, et al.: Fruits and vegetables and prostate cancer: no association among 1104 cases in a prospective study of 130544 men in the European Prospective Investigation into Cancer and Nutrition (EPIC). Int J Cancer 109 (1): 119-24, 2004
- Shike M, Latkany L, Riedel E, et al.: Lack of effect of a low-fat, high-fruit, -vegetable, and -fiber diet on serum prostate-specific antigen of men without prostate cancer: results from a randomized trial. J Clin Oncol 20 (17): 3592-8, 2002.
- BJU-2016-1793.R2 – The Men’s Eating and Living (MEAL) Study (CALGB 70807 [Alliance]): Recruitment Feasibility and Baseline Demographics of a Randomized Trial of Diet in Men on Active Surveillance for Prostate Cancer
- Fleshner NE, Lucia MS, Egerdie B, et al. Dutasteride in localised prostate cancer management: the REDEEM randomised, double-blind, placebo-controlled trial. 2012;379(9821):1103-1111.
- Cooperberg MR, Carroll PR, Klotz L: Active surveillance for prostate cancer: progress and promise. J Clin Oncol 29 (27): 3669-76, 2011. [PubMed]
- Lurdes Y.T. Inoue, Bruce J. Trock, Alan W. Partin, H. Ballentine Carter, Ruth Etzioni Modeling Grade Progression In An Active Surveillance Study Stat Med. Author manuscript; available in PMC 2015 Mar 15. Published in final edited form as: Stat Med. 2014 Mar 15; 33(6): 930–939.
Article of the Week: Early surgical outcomes and oncological results of RAPN
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Early surgical outcomes and oncological results of robot-assisted partial nephrectomy: a multicentre study
Abstract
Objective
To describe a multicentre experience of robot-assisted partial nephrectomy (RAPN) in northern England, with focus on early surgical outcomes and oncological results.
Patients and Methods
All consecutive patients undergoing RAPN at four tertiary referral centres in northern England in the period 2012–2015 were included for analysis. RAPN was performed via a transperitoneal approach using a standardized technique. Prospective data collection was performed to capture preoperative characteristics (including R.E.N.A.L. nephrometry score), and peri-operative and postoperative data, including renal function. Correlations between warm ischaemia time (WIT), positive surgical margin (PSM) rate, complication rates, R.E.N.A.L. nephrometry scores and learning curve were assessed using univariate and multivariate analyses.
Results
A total of 250 patients (mean age 58.1 ± 13 years, mean ± sd body mass index 27.3 ± 7 kg/m2) were included, with a median (range) follow-up of 12 (3–36) months. The mean ± sd tumour size was 30.6 ± 10 mm, mean R.E.N.A.L. nephrometry score was 6.1 ± 2 and 55% of tumours were left-sided. Mean ± sd operating console time was 141 ± 38 min, WIT 16.7 ± 8 min and estimated blood loss 205 ± 145 mL. There were five conversions (2%) to open/radical nephrectomy. The overall complication rate was 16.4% (Clavien I, 1.6%; Clavien II, 8.8%; Clavien III, 6%; Clavien IV/V; 0%). Pathologically, 82.4% of tumours were malignant and the overall PSM rate was 7.3%. The mean ± sd preoperative and immediate postoperative estimated glomerular filtration rates were 92.8 ± 27 and 80.8 ± 27 mL/min/1.73 m2, respectively (P = 0.001). In all, 66% of patients remained in the same chronic kidney disease category postoperatively, and none of the patients required dialysis during the study period. ‘Trifecta’ (defined as WIT < 25 min, negative surgical margin status and no peri-operative complications) was achieved in 68.4% of patients overall, but improved with surgeon experience. PSM status and long WIT were significantly associated with early learning curve.
Conclusion
This is the largest multicentre RAPN study in the UK. Initial results show that RAPN is safe and can be performed with minimal morbidity. Early oncological outcomes and renal function preservation data are encouraging.
Editorial: From Novick to the NHS – the evolution of minimally-invasive NSS
The publication in this issue of the BJUI by Veeratterapillay et al. [1] of a UK multicentre study in a community setting marks a watershed in the availability and quality of minimally invasive nephron-sparing surgery (NSS) for renal cancer. Such a turning point was predicted almost 17 years ago by Novick [2] when he wrote, ‘minimally invasive modalities of tumour resection or destruction should be reserved for highly select patients and awaits improvements in technology, standardization of technique and long-term outcomes data before they may be completely integrated options’. It appears now that robot-assisted surgery provides such a platform. The present study [1] describes the outcomes of patients treated with robot-assisted partial nephrectomy (RAPN) at four centres in Northern England, and shows very good outcomes within their first 250 cases.
The benefits of NSS have been well described. Indeed, excellent outcomes for PN were described over 20 years ago in carefully selected cases, with benefits including reduced incidence of renal insufficiency compared to radical nephrectomy, which until that time had been viewed as the ‘gold-standard’ for patients with RCC [3]. However, the popularity of PN for small renal masses appeared to decline with the advent of laparoscopy. It became apparent that a minimally invasive approach to radical nephrectomy had the advantage of improved recovery, reduced blood loss with equal cancer control to open nephrectomy [4]. Notwithstanding absolute and relative indications for PN, given the choice between an open PN and a laparoscopic radical nephrectomy, the balance for patients with an elective indication for PN was tipped in favour of a minimally invasive yet radical approach [5]. Techniques for PN were in their infancy, and even in the leading high-volume centres outcomes, including warm ischaemia time (WIT) and positive surgical margin (PSM) rate, failed to match those of open surgery [6].
Fast forward to 2017 with the increasing use of robot-assisted urological surgery carrying the advantages of three-dimensional vision, wristed movement and integrated real-time intraoperative imaging, especially beneficial for procedures such as PN where quick and accurate suturing are essential for a successful outcome. Veeratterapillay et al. [1] present a series of 250 patients from centres in the UK, in which each performs <50 RAPN procedures/year, yet the authors present favourable outcomes overall, with a PSM rate of 7.3%, major complications in 6% and trifecta in 68.4%. An impressive learning curve is seen with improving outcomes over the series, such that in the final 50 cases a trifecta (WIT <25 min, negative surgical margin and absence of complications) was achieved in 82% of cases, with a PSM rate of 2% despite increasing complex nephrometry scores, which compares favourably with larger series from internationally renowned centres [6].
So then, with the results of the present study [1], can we say that Novick’s requirements have been met, and that minimally invasive NSS is now a ‘completely integrated option’? Certainly, with the widespread adoption of robot-assisted surgery, high-quality outcomes are within the grasp of centres other than elite academic institutions. As techniques develop and experience grows robot-assisted surgery can be increasingly offered, even for resection of more complex tumours.
To ensure that minimally invasive NSS is delivered to the highest standards, it will be necessary for providers to ensure both quality assurance and quality control in their processes. The learning curve needs to be minimised with structured teaching and mentoring, and the use of adjuncts such as intraoperative ultrasonography or fluorescence should be a routine part of care.
Centres offering this technique should be mindful of the well documented volume–outcome relationship that appears to be ubiquitous among complex surgical procedures. If centres are performing less than an optimum number of cases, they may consider affiliating themselves with other such centres in networks and forming a joint clinical governance programme, as has been described for robot-assisted radical prostatectomy and which has shown demonstrable improvements in outcomes.
Finally, auditing and reporting of outcomes remains the cornerstone of quality assurance as shown by the introduction of the BAUS complex surgery audit, which is intended to drive standards of care forward. Publications such as that of Veeratterapillay et al. [1] greatly assist in documenting the progress of new techniques and emerging technologies. Increasingly, patients expect transparency from healthcare providers, and with the necessary support processes in place, such initiatives, and the data that they produce will help to further improve the delivery of complex surgery to patients from all areas of our practice.
Article of the Week: Quality Improvement in Cystectomy Care with Enhanced Recovery (QUICCER) study
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Quality Improvement in Cystectomy Care with Enhanced Recovery (QUICCER) study
Objectives
To determine if patients managed with a cystectomy enhanced recovery pathway (CERP) have improved quality of care after radical cystectomy (RC), as defined by a decrease in length of hospital stay (LOS) without an increase in complications or readmissions compared with those not managed with CERP.
Subjects and Methods
The Quality Improvement in Cystectomy Care with Enhanced Recovery (QUICCER) study was a non-randomized quasi-experimental study. Data were collected between June 2011 and April 2015. The CERP was implemented in July 2013. The primary endpoint was LOS. Secondary endpoints were quality scores, complications and readmissions. Multivariable regression was performed. Propensity score matching was carried out to further simulate randomized clinical trial conditions. A CERP quality composite score was created and evaluated with regard to adherence to CERP elements.
Results
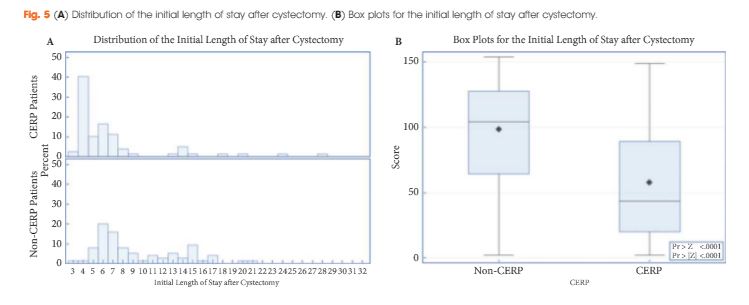
The study included 79 patients managed with CERP and 121 who were not managed with CERP. After matching, there were 75 patients in the non-CERP group. The LOS was significantly different between the groups: the median LOS was 5 and 8 days for the CERP and non-CERP group, respectively (P < 0.001). Multivariable linear regression showed that any complication was the most significant predictor of total LOS at 90 days after RC. The higher the quality composite score the shorter the LOS (P < 0.001). There was no association between CERP and a greater number of complications or readmissions.
Conclusions
Audited quality measures in the CERP are associated with a reduction in LOS with no increase in readmissions or complications. The CERP is important for the future improvement of peri-operative care for RC and provides an opportunity to improve the quality of care provided.
Article of the Week: Partial versus Radical Nephrectomy for T1 renal tumour
Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
Finally, the third post under the Article of the Week heading on the homepage will consist of additional material or media. This week we feature a video from Dr. Marios Hadjipavlou, discussing his paper.
If you only have time to read one article this week, it should be this one.
Partial versus Radical Nephrectomy for T1 renal tumours: An analysis from the British Association of Urological Surgeons Nephrectomy Audit
OBJECTIVES
To analyse and compare data from the British Association of Urological Surgeons Nephrectomy Audit for perioperative outcomes of partial (PN) and radical nephrectomy (RN) for T1 renal tumours.
PATIENTS AND METHODS
UK consultants were invited to submit data on all patients undergoing nephrectomy between 1 January and 31 December 2012 to a nationally established database using a standard pro forma. Analysis was made on patient demographics, operative technique, and perioperative data/outcome between PN and RN for T1 tumours.
RESULTS
Overall, data from 6 042 nephrectomies were reported of which 1 768 were performed for T1 renal tumours. Of these, 1 082 (61.2%) were RNs and 686 (38.8%) were PNs. The mean age of patients undergoing PN was lower (PN 59 years vs RN 64 years; P < 0.001) and so was the WHO performance score (PN 0.4 vs RN 0.7; P < 0.001). PN for the treatment of T1a tumours (≤4 cm) accounted for 55.6% of procedures, of which 43.9% were performed using a minimally invasive technique. For T1b tumours (4–7 cm), 18.9% of patients underwent PN, in 33.3% of which a minimally invasive technique was adopted. The vast majority of RNs for T1 tumours were performed using a minimally invasive technique (90.3%). Of the laparoscopic PNs, 30.5% were robot-assisted. There was no significant difference in overall intraoperative complications between the RN and PN groups (4% vs 4.3%; P = 0.79). However, PN accounted for a higher overall postoperative complications rate (RN 11.3% vs PN 17.6%; P < 0.001). RN was associated with a markedly reduced risk of severe surgical complications (Clavien Dindo classification grade ≥3) compared with PN even after adjusting for technique (odds ratio 0.30; P = 0.002). Operation time between RN and PN was comparable (141 vs 145 min; P = 0.25). Blood loss was less in the RN group (mean for RN 165 vs PN 323 mL; P < 0.001); however, transfusion rates were similar (3.2% vs 2.6%; P = 0.47). RN was associated with a shorter length of stay (median 4 vs 5 days; P < 0.001). A direct comparison between robot-assisted and laparoscopic PN showed no significant differences in operation time, blood loss, warm ischaemia time, and intraoperative and postoperative complications.
CONCLUSIONS
PN was the method of choice for treatment of T1a tumours whereas RN was preferred for T1b tumours. Minimally invasive techniques have been widely adopted for RN but not for PN. Despite the advances in surgical technique, a substantial risk of postoperative complications remains with PN.
Editorial: Minimally invasive surgery or nephron preservation for small renal tumours?
In the present issue of BJUI, there is an important study by Hadjipavlou et al. [1], summarizing radical (RN) and partial nephrectomy (PN) practice in the UK in 2012. Specifically, the authors reported the outcomes of ~1 800 patients undergoing either RN or PN for clinical T1 renal masses. Approximately 55% of the patients with cT1a tumours underwent PN, of whom 44% underwent minimally invasive PN. Conversely, in the cohort of patients with cT1b tumours, only ~19% received PN, of whom 33% underwent a minimally invasive procedure. Notably, whereas operating time, transfusion rate and the risk of intraoperative complications was similar for RN and PN, postoperative complications were approximately three times more common in patients who underwent PN, after adjusting for covariates. A sub-analysis comparing robot-assisted and laparoscopic PN failed to show any difference in peri-operative outcomes [1].
The study is important for several reasons. Firstly, it shows a fairly high adoption of PN for cT1a tumours. Although PN is recommended as the standard treatment for small renal masses [2], population-based studies have shown that there has been limited adoption of PN outside referral centres [3, 4], especially in the USA. Conversely, the present data from UK show more encouraging results, maximizing the benefit of nephron preservation; however, although PN might be more challenging in cT1b tumours and the available evidence in favour of PN in such a setting is less compelling, the adoption of PN was lower in such tumours. Efforts should be made to popularize such an approach whenever feasible.
Secondly, the study showed that a minority of the PN procedures were performed with a minimally invasive approach. Although we can agree that nephron preservation is more important than a minimally invasive approach in the long term for most patients, an increasing number of publications and growing clinical experience suggest that laparoscopic, and, above all, robot-assisted PN could represent the ideal solution. Although the number of minimally invasive PNs should increase with increased diffusion of DaVinci platforms, major efforts should be made to expand the number of patients in whom the morbidity of the traditional open PN approach can be avoided. In this context, regionalization of care for PN, as for other major oncological procedures, could be an excellent solution.
Thirdly, the significant rise in the risk of postoperative complications observed after PN could allow better selection of patients to undergo either PN or RN. For example, where surgery is indicated, frail comorbid patients, in whom the risk of perioperative complications should be minimized and who would benefit less from nephron preservation, could be better treated by laparoscopic RN or, probably, robot-assisted PN as performed by very experienced surgeons.
Finally, the study failed to show major differences between laparoscopic and robot-assisted PN. Although this finding is in line with data from systematic reviews of the literature [5], the present data from a large cohort of surgeons are more solid. The lack of data on patient selection, previous laparoscopic and robot-assisted surgery, annual surgical volume and tumour characteristics according to nephrometry scores, however, does not allow us to draw definitive conclusions on the issue. In our opinion, robot-assisted surgery might offer major significant benefits during PN in terms of quicker and more accurate tumour dissection, improved renorrhaphy with consequent shorter ischaemia time, lower risk of complications and a shorter learning curve as compared with pure laparoscopic PN.
Unfortunately, no analysis stratified by centre and/or surgeon volume was provided in the present paper. As with other major surgical procedures, some studies suggest that case volume may have a major impact on outcome [6]. It would have been interesting to see such a relationship analysed in the present cohort involving almost 300 surgeons from more than 100 institutions. Despite the large number of cases analysed, however, it is likely that these data depict the outcomes of RN and PN in a low-volume setting (an average of approximately six cases per year in total).
Finally, alternative approaches such as percutaneous or laparoscopic cryoablation are gaining popularity for the treatment of small renal masses in selected cases [2]. Although long-term oncological outcomes of such procedures are lacking, the available evidence suggests good short-term efficacy and safety for cryoablation in patients with small renal masses. The presence of data on such treatments to compare with the surgery results reported in the present cohort would also have been of interest.